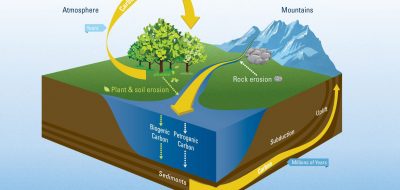
Alkalinity refers to water’s ability to neutralize acid. When alkaline molecules react with carbon dioxide, it converts to a form that can’t readily return to the atmosphere as carbon dioxide gas. Illustration by Natalie Renier, ©Woods Hole Oceanographic Institution
What is ocean alkalinity?
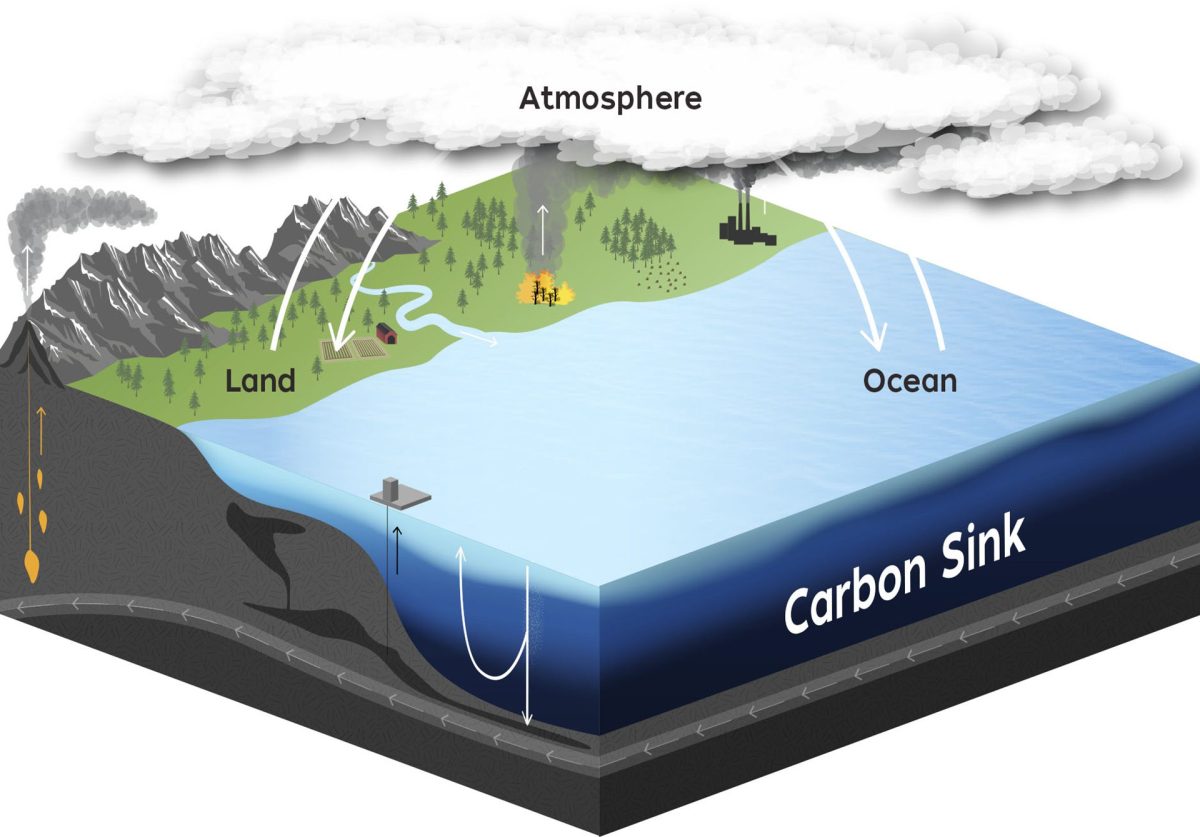
Over the past century, rapidly increasing levels of atmospheric carbon dioxide have been held in check by our oceans, which absorb the gas from the air. This process happens at the water’s surface, as carbon dioxide moves between water and air in an attempt to achieve equilibrium. Rising levels of atmospheric carbon dioxide have shifted that equilibrium, causing more of the gas to move into the ocean. As carbon dioxide enters the water, some reacts with molecules in seawater or is used by photosynthetic organisms. Some is pumped to deeper layers of water through currents or biological processes. And some reacts with alkaline molecules weathered from rock or ancient shells.
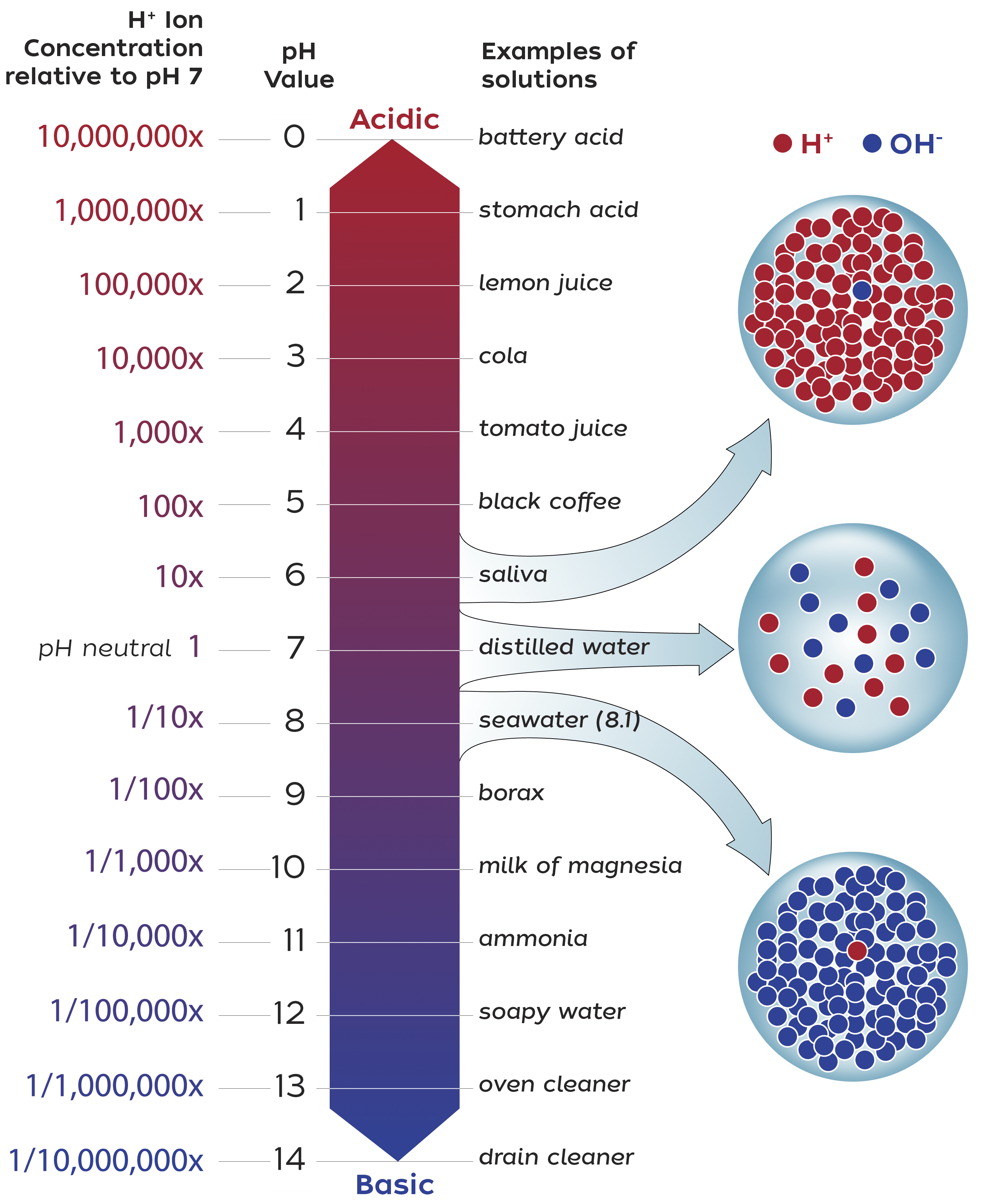
Alkalinity refers to water’s ability to neutralize acid. We hear a lot about the problem of ocean acidification, which is driven by the reaction of carbon dioxide and water. This reaction releases hydrogen ions that make the water more acidic, lowering its pH. These ions readily react with other molecules, including those found in sea shells and coral skeletons, presenting a grave problem for shelled marine life. Alkaline molecules, on the other hand, are basic—they react with those hydrogen ions, locking them into new, more stable molecules. This raises the pH of the water. Alkaline molecules also react with carbon dioxide, converting it to a form that can’t readily return to the atmosphere as carbon dioxide gas.
Why is it important for climate change?
As carbon dioxide reacts with alkaline molecules, the concentration of carbon dioxide gas in the water decreases. This disrupts the equilibrium between air and water, drawing more of the gas into the water. This natural process has been happening for billions of years, helping to regulate the ocean’s pH. More recently, as humans have produced large amounts of carbon dioxide, oceans have removed much of that gas from the atmosphere. It’s thanks to this process that global temperatures have risen more slowly than they otherwise would.
Reaction of carbon dioxide with alkaline molecules occurs naturally, but because those molecules are weathered from rock or ancient shells, it happens on geologic timescales. Weathering is a slow process, and the number of alkaline molecules in the oceans is becoming limited. Researchers are now exploring the possibility of speeding the process by adding crushed minerals to beaches or directly to the ocean via ships.
The addition of alkaline minerals has the potential to remove gigatons of carbon each year. Although global leaders are working toward a carbon neutral future, researchers estimate that the only way to keep global temperature increases below 1.5 degrees Celsius and prevent a climate catastrophe is to remove carbon dioxide that’s already in the atmosphere. Ocean alkalinity enhancement is one of several promising tools that could help achieve this goal.
How are scientists studying ocean alkalinity enhancement?
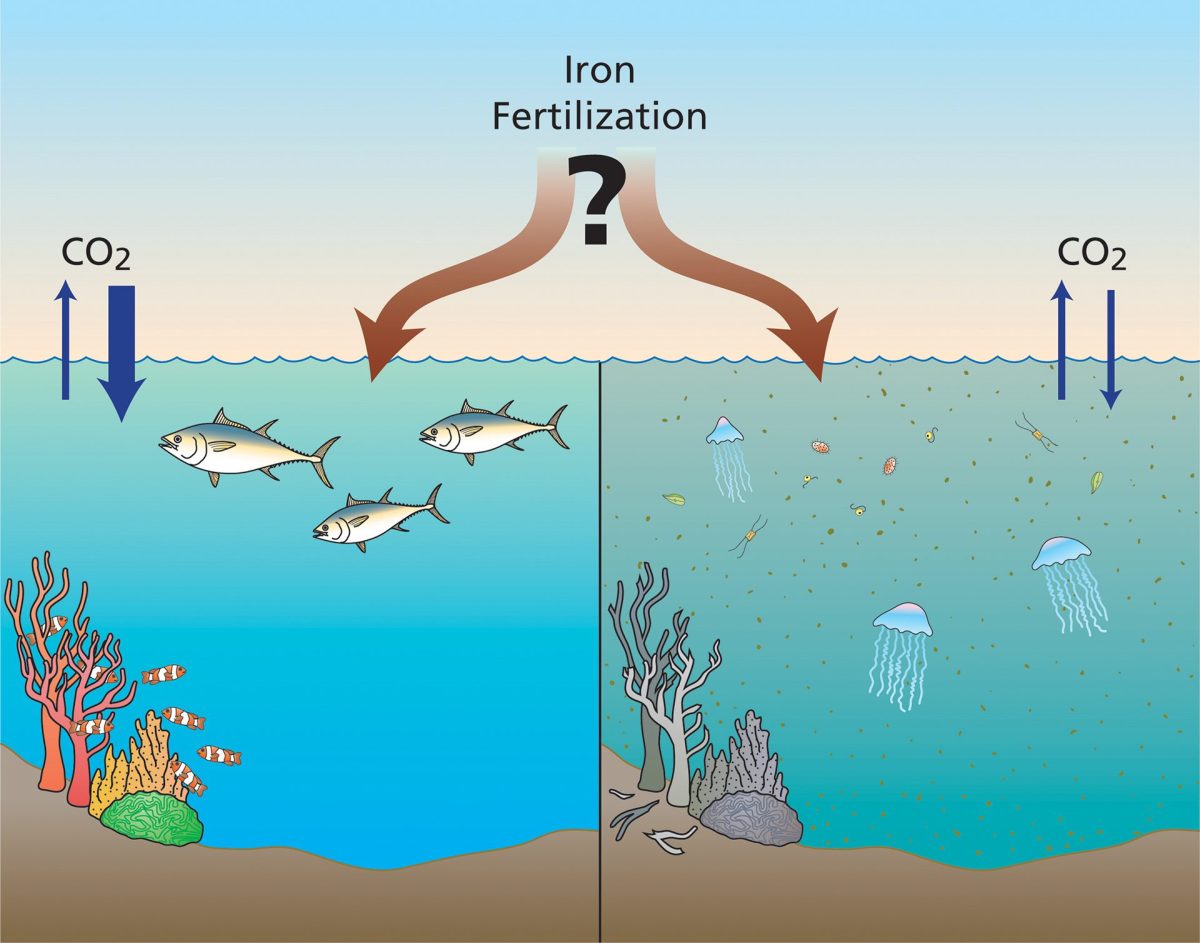
Alkaline molecules are a natural part of the ocean and its processes, but they are produced slowly—over hundreds to thousands of years—as rock on land is weathered and the minerals are washed into rivers and eventually out to sea. Enhancing the ocean’s alkalinity on a much faster time scale (years to decades) could potentially harm marine ecosystems, so scientists are working to determine what kinds of impacts might arise from such efforts before undertaking large-scale alkalinity enhancement projects.
WHOI researchers are investigating the possible impacts of increased alkalinity on phytoplankton. These tiny photosynthetic organisms form the base of the marine food web, and changes in their ability to survive could have wide-ranging impacts on the rest of the ocean. Ocean acidification has been damaging coral skeletons and shells, including those of some phytoplankton, so alkalinity enhancement has the potential to reverse ongoing damage caused by carbon dioxide driven changes in pH. But before it can be used at large scales, it’s essential to determine whether such efforts will be safe for marine ecosystems.
Other researchers are using computer models to better understand the environmental factors that could impact efforts to enhance alkalinity. Ocean currents, water temperature, proximity to land, and time of year all affect how easily alkaline minerals can remove carbon dioxide from the atmosphere. How rapidly carbon dioxide moves from air into water also plays a role. These models suggest there could be profound differences between regions of the ocean in their ability to absorb carbon dioxide. Because the oceans are so vast, identifying specific locations where mineral enhancement would be most effective—and least damaging—is essential.
Bach, L.T. et al. CO2 removal with enhanced weathering and ocean alkalinity enhancement: Potential risks and co-benefits for marine pelagic ecosystems. Frontiers in Climate. October 11, 2019. doi: 10.3389/fclim.2019.0007.
Burt, D.J. et al. The sensitivity of the marine carbonate system to regional ocean alkalinity enhancement. Frontiers in Climate. July 9, 2021. doi: 10.3389/fclim.2021.624075.
Castañón, Laura. An ocean of opportunity. Oceanus. December 7, 2021. https://www.whoi.edu/oceanus/feature/an-ocean-of-opportunity/
Doney, S.C. et al. A research strategy for ocean-based carbon dioxide removal and sequestration. Consensus Study Report Highlights. December 2021. https://www.nap.edu/resource/26278/Ocean_CDR_2021.pdf
Federer, A. et al. Assessing the influence of ocean alkalinity enhancement on a coastal phtyoplankton community. Biogeosciences. Preprint. 2022. doi: 10.5194/bg-2022-17.
Lubofsky, Evan. The ocean has a serious case of heartburn. Is relief on the way? Oceanus. July 1, 2021. https://www.whoi.edu/oceanus/feature/ocean-alkalinity/