An ocean of opportunity
Ocean experts explore the potential risks and rewards of ocean-based solutions to climate change
 This article printed in Oceanus Fall 2021
This article printed in Oceanus Fall 2021
Estimated reading time: 23 minutes
If you shine a light in the deep ocean, you might imagine yourself in a Hollywood version of outer space. The surrounding darkness seems endless. Oddly luminous creatures swim in and out of view. And everywhere the light touches, it bounces off millions of tiny flecks of detritus-a drifting starscape of carbon.
The ocean is a vast repository for carbon. Over the last century, it has absorbed nearly a third of the carbon dioxide that humans have pumped into the atmosphere through the burning of fossil fuels. This carbon ends up dissolved into the water, captured by organisms, folded into coastal sediments, or buried in the deep sea.
But even with all the carbon the ocean removes from the atmosphere, our planet is on track to exceed a global 1.5°C temperature increase within the next 20 years unless we take immediate measures to reverse this trend, according to the 2021 report from the Intergovernmental Panel on Climate Change. Drastically reducing emissions is essential. But at this point, we need to do more than just cut our reliance on fossil fuels. We need to find new ways to remove and store the excess carbon dioxide we've already produced.
The ocean, which covers more than 70% of our planet, may be able to help. Researchers and commercial interests are actively looking for ways to intentionally enhance the ocean's natural processes to sequester additional carbon. Some of these measures may also begin to address other ocean issues such as acidification or nutrient runoff. But there is no silver bullet for climate change; each carbon storage method will have significant trade-offs and risks.
How, then, can we ensure that in our rush to solve the climate crisis, we do not inadvertently destroy critical ocean ecosystems and the global systems that rely on them?

“Marine snow” is a catchall term for organic detritus and fecal pellets that clump together to form millimeter-long flakes as they fall through the water column. This image was captured at 55 meters (180 feet) in Monterey Bay, California, by a light-scattering optical device that also counts and estimates the size of particles. (Photo courtesy of Susumu Honjo, © Woods Hole Oceanographic Institution)
"Ocean-based strategies should not be enacted without a careful assessment of their long-term viability and potential impacts to the marine environment," wrote Woods Hole Oceanographic Institution (WHOI) President and Director Peter de Menocal and WHOI Deputy Director Rick Murray in a recent Boston Globe op-ed piece. "There must be in place comprehensive, scalable, and long-term coastal and ocean monitoring systems that track ocean health and vital signs, to ensure the welfare of future generations."
"The more information you have, the better you can plan and that's some of what Woods Hole does: We run experiments, gather data, and try to learn and understand so that we can help people plan and adapt."
—Carol Anne Clayson, WHOI's director of research strategies
Before humans throttle up the ocean's carbon storage capabilities, we need to understand the impacts on the ocean, the efficacy of these interventions, and their effects on sea life and human communities. We need the scientific knowledge to make informed decisions, and systems in place to spot when things go awry. Researchers at WHOI and other institutions are stepping up to tackle these urgent challenges.
"The more information you have, the better you can plan," says Carol Anne Clayson, WHOI's director of research strategies. "And that's some of what Woods Hole does: We run experiments, gather data, and try to learn and understand so that we can help people plan and adapt."
How the ocean stores carbon
The surface ocean and the air above it are constantly exchanging gases, trying to reach an equilibrium. When carbon dioxide is more plentiful in the atmosphere than in the surface waters, it diffuses into the ocean. There, physical and biological processes work to pump some of the carbon into deeper waters, making room for more absorption at the surface.
Some dissolved carbon dioxide bonds with molecules weathered from rocks or ancient shells, locking it into a new, complex form that cannot easily escape back into the atmosphere. Other carbon is dragged to the depths by major ocean currents, which pull warm surface waters laden with carbon dioxide toward the poles, where they cool and sink. Once carbon has reached the deep ocean, it can remain there for hundreds to thousands of years.
On the biological side, marine algae play a key role, using dissolved carbon dioxide to conduct photosynthesis. When those phytoplankton and other microorganisms die, they too sink to deeper waters.
"If you don’t do the responsible oceanography to look at consequences, it’s pretty easy to make a big mess."
—WHOI geochemist Ken Buesseler
"The biological carbon pump sends on the order of five to 12 gigatons of carbon per year out of the surface," says Ken Buesseler, a geochemist at WHOI. The combined mass of every human on the planet is about half a gigaton. "But it doesn't all get to the bottom."
Researchers estimate that only about 10% of marine snow, as it's known, makes it more than 1,000 meters deep. Organisms in the ocean twilight zone-a shadowy midwater region of the ocean with too little light for photosynthesis-consume most of this falling detritus. And when animals eat, they convert organic carbon back into carbon dioxide as they breathe out.
These animals act like a short circuit, rerouting sinking carbon, Buesseler says. "If carbon doesn't get deep enough, the water just mixes it back up to the surface in the wintertime with a storm or with natural ocean currents." Carbon at the surface can seep back into the atmosphere.
The amount of carbon reaching the deep ocean varies from place to place depending on the depth sunlight can penetrate, the availability of nutrients, and fluctuations in currents and temperature. Carbon storage solutions intended to mimic or enhance these natural cycles will need to account for the same variables.
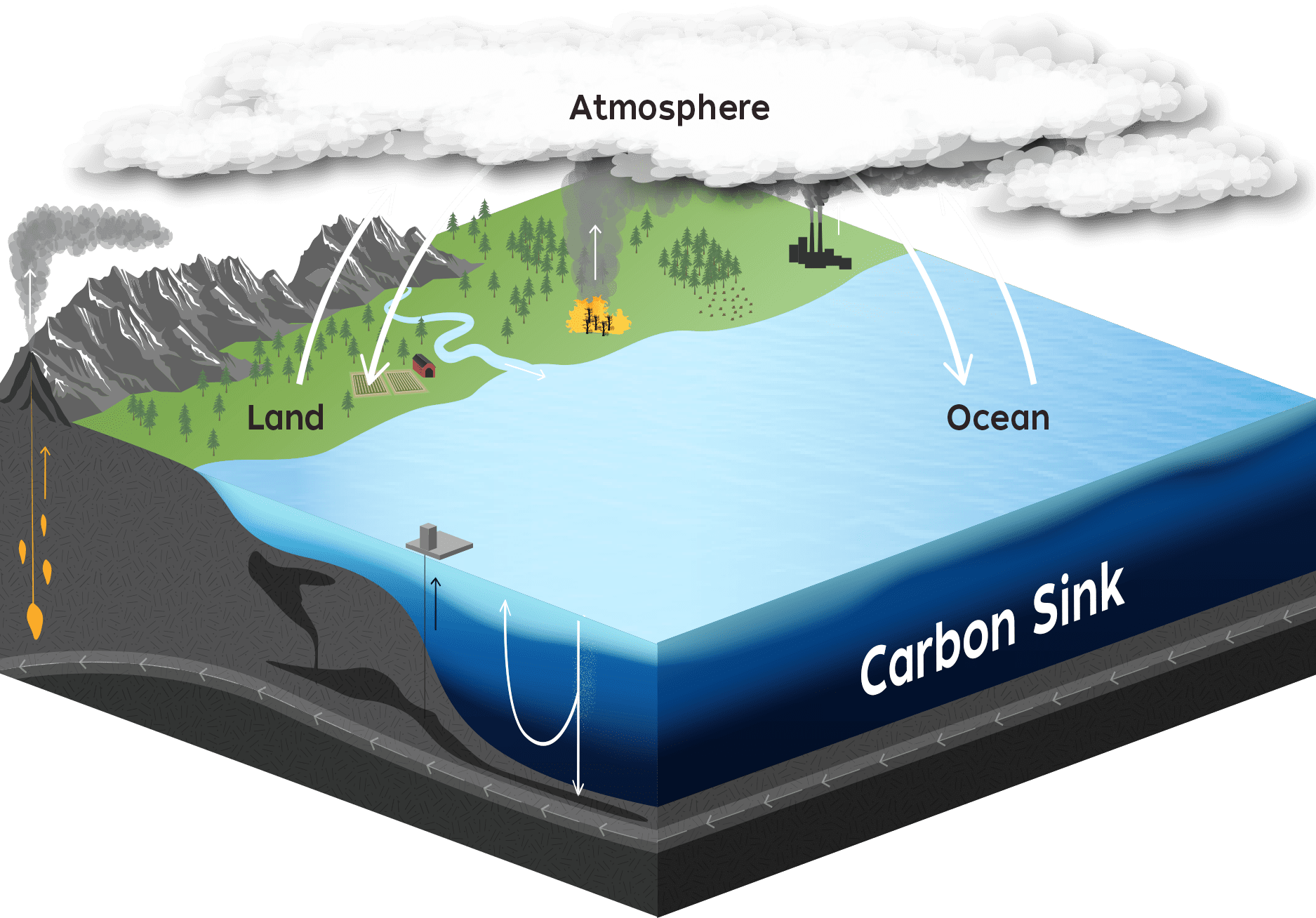
In various forms, carbon is continuously exchanged between Earth's atmosphere, land, and water-an essential cycle for life and regulating the planet's climate. Atmospheric carbon dioxide readily dissolves in the ocean's surface waters, where some of it is taken up by living organisms or sequestered in deep-sea rocks and sediments. (Illustration by Natalie Renier, WHOI Creative, © Woods Hole Oceanographic Institution)
Fertilizing the ocean
In 1988, oceanographer John Martin stood in WHOI's Redfield Auditorium, discussing his theory that adding iron to the ocean would cause phytoplankton blooms, which could use up excess carbon dioxide in the water. He concluded his lecture declaring, "Give me half a tanker of iron, and I will give you an ice age."
The oft-quoted line was a joke (Martin later described it as being delivered in his "best Dr. Strangelove accent"), but the possibility of ramping up the ocean's productivity to undo human-induced changes caught the attention of both researchers and commercial entities at the time.
While iron is the fourth-most-abundant element in Earth's crust, it is extremely scarce in the ocean. Most iron ore is locked up in rock formations and becomes available only as those rocks are weathered by the elements. Rivers and strong winds pick up trace amounts of iron dust and carry it to the sea, where phytoplankton use it for photosynthesis.
"All plants need some amount of iron and, since it comes from things like dust, it's not evenly distributed over our water," says Buesseler, who conducted a series of small-scale iron fertilization experiments in the open ocean in the '90s and early 2000s. Areas of the ocean that are closer to large landmasses are more likely to have a consistent supply of iron. "The southern half of the globe has far fewer landmasses than the northern part. So, particularly in the Southern Ocean, there are huge regions that if you experimentally add a little more iron, things grow faster."
During the experiments, the infusion of iron successfully caused localized plankton blooms, but further research was needed to determine how much, if any, of the extra organic material would make it to the deep sea. Instead of drifting to the depths in a snowstorm of dead cells, Buesseler and his colleagues realized there was a chance that the sudden excess of phytoplankton would cause a corresponding boom in other species up the food chain. Much of the organic carbon, the researchers thought, might be consumed near the surface and converted back to carbon dioxide.
"We need to work together, we need to compile and combine all the resources out there-observation and measurement data, experimental data, and modeling data-to make predictions about how each of these carbon dioxide removal methods is going to impact the ocean ecosystem health over the long term."
—WHOI biogeochemist Heather Kim
The researchers also wanted to better understand potential side effects of turbocharging the biological pump: Plankton blooms could deplete surface waters of nutrients that other species depend on, and the influx of decaying organic material might use up oxygen and create dead zones. But before they were able to thoroughly investigate both the effectiveness and possible harms of iron fertilization, some commercial ventures were already springing up to capitalize on the idea. In response to growing fears that unregulated iron fertilization would irreversibly damage ocean ecosystems, most research in this area stopped in its tracks.
"A lot of this anxiety was well deserved, because people were just going off and making these claims and trying to commercialize it before it was ready," Buesseler says. "If you don't do the responsible oceanography to look at consequences, it's pretty easy to make a big mess."
But the ocean is already changing rapidly, and ocean fertilization, whether with iron or other nutrients, has resurfaced as one possibility in a suite of options to try to increase carbon storage.
WHOI biogeochemist Heather Kim is working on a proposal to build a numerical modeling framework to assess the potential long-term effects of ocean iron fertilization. Kim, who specializes in modeling the interaction between physical, chemical, and biological aspects of the ocean, intends to evaluate how effective iron could be at mitigating climate change, as well as the potential environmental risks and benefits. Her work will rely on decades of fundamental research into ocean processes, as well as the prior iron fertilization experiments.
"We need to work together," Kim says. "We need to compile and combine all the resources out there-observation and measurement data, experimental data, and modeling data-to make predictions about how each of these carbon dioxide removal methods is going to impact the ocean ecosystem health over the long term.

WHOI researchers tested the effects of adding alkalinity to seawater in a series of experiments at sea. The large blue tanks shown here were kept at a sea surface temperature and light level that would be found at 15 meters (49 feet). Researchers then filtered the water through circular disks (in the foreground) to sample the remaining particulate material after incubation. (Photo by Ashley Maloney, Princeton University)
Just add alkalinity
Over geologic timescales, alkaline molecules weathered from rocks or released by the dissolving shells of long-dead microorganisms help the ocean lock away a significant amount of carbon and play an important role in regulating the carbon dioxide levels in our atmosphere. But since we don't have millions of years to address climate change, some scientists are investigating the possibility of boosting the ocean's alkalinity to mimic the effects of rock weathering over a much shorter time scale.
Alkaline molecules react with dissolved carbon dioxide to create carbonate and bicarbonate, forms of carbon that don't diffuse easily back into the atmosphere. At the sea surface, this reaction lowers the amount of dissolved carbon dioxide relative to the adjacent atmosphere, which encourages more carbon dioxide to diffuse into the ocean. Essentially, ocean alkalinity helps make space for more carbon storage.
But alkalinity from the slow and steady weathering of rocks can't keep pace with how rapidly we are producing carbon dioxide.
"The ocean is going to be less and less able to take up more CO2 because it's basically becoming saturated," says Adam Subhas, a marine chemist at WHOI. "By adding alkalinity, we could restore the ability of the ocean to continue to soak up more CO2."
"The ocean has soaked up hundreds, if not thousands, of gigatons of carbon before. Every option needs to be on the table, and needs to be researched seriously, because of the scale of the problem."
—WHOI marine chemist Adam Subhas
There are several potential ways to add alkalinity to the ocean. One proposal is to seed beaches with finely crushed olivine, an igneous rock that weathers relatively quickly, and let wave motion dissolve it into the sea. Other proposals involve using electric currents to induce alkalinity or scattering alkaline minerals from vessels already crossing the ocean.
"Each one of those has very different engineering challenges, costs, and risks associated with them," Subhas says. "But from my perspective, the big question is: Does adding alkalinity to the ocean do anything to marine ecosystems?"
When carbon dioxide dissolves into the ocean, it combines with seawater to form carbonic acid. The acid rapidly breaks down, releasing hydrogen ions. This process lowers the pH of the ocean and increases its acidity. In a stable climate system, alkalinity provides a buffer by snatching up the excess hydrogen to form bicarbonate, thereby stabilizing the pH.
As human-made sources of carbon dioxide have outpaced natural sources of alkalinity, the ocean has grown increasingly acidic. Adding alkalinity to the ocean may counteract ocean acidification, in addition to helping with our carbon problems.
But the rapid addition of anything to the ocean has the potential for unintended consequences, even if it is mimicking a natural process. Subhas and his colleagues have been running experiments to investigate potential impacts. Last year, the group collected samples from two sites-one near the Bahamas and one in the central North Atlantic Ocean-to look for changes in how phytoplankton fix carbon or certain species build shells when alkalinity is added to the water.
"We're starting to learn that we're not going to be able to bring things back to exactly the way they were," Subhas says. "There are going to be other effects, and the only way to understand them is to actually look and see what those effects are at a small scale, then build up to larger scales as people are ramping up on the technology side."
But Subhas is optimistic that alkalinity enhancement could be one piece of a larger climate solution.
"The ocean has soaked up hundreds, if not thousands, of gigatons of carbon before," Subhas says. "Every option needs to be on the table, and needs to be researched seriously, because of the scale of the problem."

A diver harvests kelp at an offshore aquaculture farm. WHOI researchers are developing techniques to expand kelp cultivation, which has the potential to provide low-impact nutrition, renewable energy, and carbon storage. (Photo by Paul Caiger, © Woods Hole Oceanographic Institution
Seaweed storage
Forests on land are important carbon sinks, but fires and logging can rapidly send decades of stored carbon back into the atmosphere. So, what about forests in the ocean? Large-scale seaweed farms could potentially soak up carbon and store it in much the same way.
In recent years, seaweed aquaculture has been proposed as a replacement for land-grown biofuels and animal feed. As a crop, seaweed has several advantages: It doesn't require that land be cleared for farming; it doesn't need additional water or nutrients beyond what is already available in the ocean; and it can grow extremely quickly-in some cases more than a foot per day.
These same characteristics make seaweed a good candidate for sequestering carbon. Seaweed could be grown at large scales and then sunk en masse, locking away carbon in the deep ocean.
The biggest problem right now is scale. Most seaweed farms in the U.S. are small, between one and 10 acres, and grow seaweed primarily for people to eat. These farms can be tucked into coastal areas that are protected from strong winds and waves. But growing enough seaweed to make a dent in our global carbon dioxide problem is going to take much larger operations.
Scott Lindell, an aquaculture research specialist at WHOI, is working on a project to develop new techniques and technologies for large-scale farms to operate efficiently, potentially in open-water environments. He and his colleagues have commercial pilot farms off the coasts of New England, southwestern Puerto Rico, and Kodiak, Alaska, where they are prototyping their technology.
"It's a big step up from what we're producing for human food now," Lindell says.
Lindell specializes in breeding seaweed for easier cultivation, similar to how we have bred farm crops for higher yields, faster harvest, or better taste. The project, which is funded by the U.S. Department of Energy, is aimed at turning seaweed into biofuel, but Lindell says the breeding technology could easily be tailored to optimize seaweed for other climate-friendly uses, such as biodegradable plastics.
"I think the first and most obvious thing to do is look at ways of using this low-input, low-carbon-footprint biomass to replace fossil fuels and the usual problems that are producing CO2 emissions," Lindell says. "When we've satiated all our food and packaging needs, then the next step would be to use seaweed to store carbon."
"I think the first and most obvious thing to do is look at ways of using this low-input, low-carbon-footprint biomass to replace fossil fuels and the usual problems that are producing CO2 emissions."
—WHOI aquaculture specialist Scott Lindell
But what species of seaweed should we use? How deep does it need to sink, and how do we make sure it gets there? How much carbon will be trapped in the deep ocean? And how will a sudden deluge of vegetation impact deep-sea environments? Lindell is working with WHOI's Tom Bell on a proposal for the Department of Energy to start answering these questions.
"On paper, it sounds great: Grow seaweed, sink it to a thousand meters," Lindell says. "But there's a lot of technical things in between to figure out."
Bell, an ecosystem ecologist, began some of this work at the University of California Santa Barbara, using a model to estimate how long carbon stored in various parts of the deep ocean would remain sequestered.
"If you sink biomass like kelp into the ocean, studies have shown it will decompose after a few years and the carbon will dissolve into the water," Bell says. "It will stay in the solution for a while, until it comes back into contact with the atmosphere."
In some cases, the model shows, that could be hundreds or even thousands of years. But frequently, carbon-saturated water makes its way to the surface much sooner, potentially releasing carbon back into the atmosphere within only 50 years.
Depending on global currents, certain areas of the ocean will be more suited to sinking carbon-laden biomass than others. But even temporary storage might be valuable if it could be done responsibly, Bell says, because it could buy us time. "Even 50 to 100 years is a good starting point. Hopefully, that will get us to the point where we can efficiently sequester carbon directly from the atmosphere with other, more permanent, technologies."

Matt Long deploys a solar-powered buoy off one of the Elizabeth Islands in southeastern Massachusetts, which will help track the flow of temperature, salinity, pH, oxygen levels, and light in areas with natural seagrass beds. This information provides necessary insight into the health of seagrass beds and their ability to take up carbon. (Photo by Laura Castañón, © Woods Hole Oceanographic Institution)
Carbon on the coast
At low tide, the water near Naushon Island off the coast of Cape Cod is warm and shallow, and more than a bit murky. Tufts of algae float by like cotton clouds as WHOI scientist Matt Long and postdoctoral researcher Jeff Coogan lower a heavy device comprising a series of vertical tubes and sensors rising from a torpedo-shaped battery pack into the water.
The researchers are studying how seagrass beds take up and store carbon around this small island near Woods Hole, as well as in several other locations around Cape Cod.
"If you don't have those seagrass systems, a big storm can come through, churn up the sediments, and all that carbon is back in the water."
—WHOI scientist Matt Long
Coastal ecosystems like seagrass beds, mangroves, and salt marshes hold an immense amount of carbon, but centuries of human activity have degraded or destroyed many of these areas. Restoring coastal ecosystems, and protecting those that remain, could help us maximize their potential for carbon storage.
In addition to absorbing carbon dioxide, seagrass and other plants often tangle up and trap other organic carbon that is moving through the ocean and deposit it in the sediment. Over time, decaying plants and organic matter build up into a carbon-rich muck, which is locked in place by root systems.
"There's a huge amount of carbon in the sediments that comes out when we lose them," says Long. "If you don't have those seagrass systems, a big storm can come through, churn up the sediments, and all that carbon is back in the water."
Using a solar-powered buoy he constructed in his basement while WHOI labs were closed for the COVID-19 pandemic, along with several underwater instruments, Long can track water temperature, salinity, light, pH, and rates of oxygen exchange in the seagrass bed. Carbon flux is challenging to measure directly in the ocean because of the chemical reactions that happen when carbon dioxide dissolves in seawater. But measuring the oxygen produced by plants during photosynthesis (which uses carbon dioxide) can provide the same information.
By tracking these measurements through different seasons and conditions, Long can start to understand how different conditions affect a seagrass bed's overall growth and metabolism.
Long says, "Once we better understand some of those processes, we can then begin to say, 'Okay, what are the conditions that we need to maintain this system right here but also restore other systems in other places?'"
Researchers estimate that we have lost 29% of seagrass beds globally in the last century. Rising ocean temperatures and light-blocking blooms of algae fueled by nutrient runoff create increasingly difficult conditions for seagrass to survive.
For areas where the water has grown too murky for seagrass to grow on the bottom, Long has an idea. He is working on designs for a seagrass-growing platform that could be suspended just below the surface. The plants could get enough light to do what they are supposed to do: pull excess nutrients out of the water, store carbon, and trap particles floating in the water. But, he stresses, the first step is to preserve the coastal ecosystems we still have. Then we can work on restoration.
"All those environments-marshes, mangroves, seagrass-they're all in global decline," Long says. "These are all pretty heavily impacted environments where people are directly interacting with the ocean. Let's not destroy the carbon stores we already have."

WHOI microbiologist Julie Huber, shown here giving a talk at a National Geographic event in 2018, is helping to build a network of deep-sea researchers and other stakeholders to help accelerate the pace of crustal ocean research. (Photo by Taylor Mickal, © National Geographic Society)
A seafloor vault for liquid carbon
Researchers are also considering bypassing the ocean's biological carbon pumping processes entirely. One proposed solution is to pump carbon dioxide straight into the crustal ocean rock underneath seafloor sediments.
Large sections of the upper ocean crust are made of volcanic basalt. These porous formations could be used as deep-sea vaults, storing liquid carbon dioxide until it converted into solid rock.
"When the carbon interacts geochemically with basalt, it can form limestone," says Andrew Fisher, a hydrogeologist at University of California Santa Cruz. "So, you can go from something that is gas or liquid and is highly mobile to something that is a rock or a mineral."
This mineralization process, which has been demonstrated in land-based basalt formations in Iceland, takes about two years-fast enough to effectively lock carbon away, but slow enough for it to diffuse into the formation (making space for more carbon dioxide). Dense ocean sediments could act as a cap, keeping the carbon from escaping before it mineralized.
"We want to partner with scientific and industrial organizations, bring the right scientific questions to the table, and help enable that research to happen.”
—WHOI deep-sea microbiologist Julie Huber
An international group of researchers led by Ocean Networks Canada is exploring how to set up a pilot study for this method in the waters off British Columbia. They are proposing to build a floating platform powered by renewable energy to scrub carbon from the atmosphere and inject it into the seafloor.
"We have the technology, most likely, to do a feasibility study now," says Fisher, who has been assisting with the work. "What's required is the will, the funding, and enough people putting their minds to it."
One of the critical questions the researchers will have to answer is how this will affect unique deep-sea environments and the creatures that live there.
"There are microbes all over the subseafloor, fixing CO2 into their biomass or just eating organic carbon, and they're having an impact on carbon cycling," says Julie Huber, a deep-sea microbiologist at WHOI. "So, what is going to happen if you inject this massive amount of CO2 into the deep ocean? What's going to happen with pH? What's going to happen with microbial activity?"
Supported by the National Science Foundation, Huber and Fisher, along with scientists at several other institutions, are building a network of deep-sea researchers and other stakeholders to help speed up the pace of crustal ocean science and answer some of these pressing questions. The Crustal Ocean Biosphere Research Accelerator, or COBRA, will help provide the information needed to make knowledgeable decisions about further uses of the deep sea.
“One of the reasons we know so little is because access to deep rocks beneath the seafloor is really challenging,” Huber says. “Drilling into ocean crust is a big job and requires a lot of special technology. So, from the COBRA perspective, we want to partner with scientific and industrial organizations, bring the right scientific questions to the table, and help enable that research to happen.”
An ocean of snow: Since the dawn of humanity and long before, the ocean has quietly absorbed carbon dioxide from the atmosphere. Creatures like this filter-feeding larvacean play a key role in the carbon storage process by taking up and excreting this molecule to deeper waters. But as industries continue to belch inordinate amounts of this greenhouse gas, a big question looms: Can the ocean do more to store carbon? Researchers are investigating the efficacy of a range of potential solutions, and what the impacts might be on marine ecosystems and animals like this larvacean. (Photo courtesy of the Mesobot Team, © Woods Hole Oceanographic Institution)
An ocean decade
This year marks the start of the United Nations' Decade of Ocean Science for Sustainable Development. It is an opportunity to bring together global collaborations to conduct "the science we need for the ocean we want." The effects of climate change are threatening coastal communities and ocean ecosystems alike-this is the perfect moment to look to the ocean for solutions.
"Responsible ocean carbon dioxide removal is intended to reduce suffering and loss from climate change; that's the goal. If we think there's an option by altering alkalinity, enhancing productivity, or some other means, we should certainly explore it."
—WHOI geochemist Ken Buesseler
Transitioning away from fossil fuels and pulling excess carbon from the atmosphere is going to require a whole portfolio of options, some of which may rely on storing carbon in the ocean. To enact any of them responsibly, we need the scientific knowledge to understand the potential benefits and mitigate the potential harms of each option.
"Responsible ocean carbon dioxide removal is intended to reduce suffering and loss from climate change; that's the goal," Buesseler says. "If we think there's an option by altering alkalinity, enhancing productivity, or some other means, we should certainly explore it."