In this section
Ocean Topics
- Climate & Weather
- How the Ocean Works
- Ocean & Human Lives
- Ocean Life
- Sustainable Ocean
- Ocean Tech

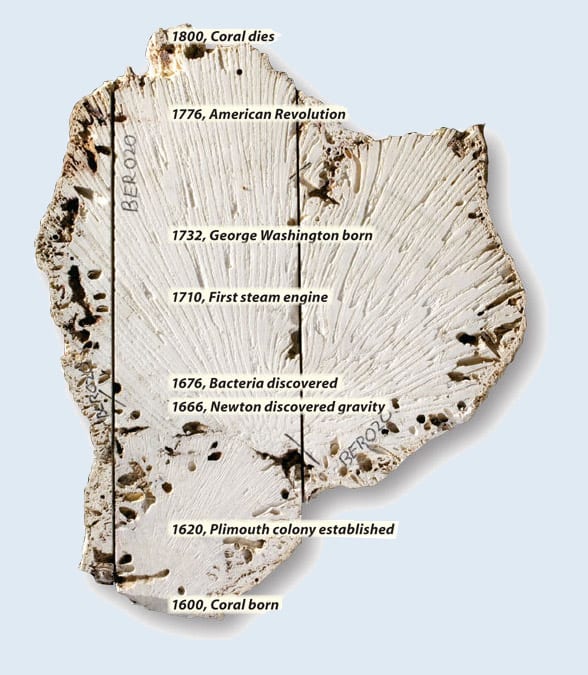
A slice through the center of a long-dead brain coral is a slicethrough human and ocean history. This 1,000-pound coral grew nearBermuda for 200 years. WHOI Research Associate Anne Cohen and fellowscientists are analyzing coral skeletons to decipher ocean temperaturesas recorded in the rock-like layers during its lifespan. (Photo by Tom Kleindinst, Woods Hole Oceanographic Institution)
Coral is a useful tool for scientists who want to understand changes in past climate, but recalling that history presents its own set of challenges.
In order to know anything about past climate from corals, we need to know their age. Ordinarily, scientists determine an object’s age by measuring the radioactive decay of some element in it. This decay occurs when an unstable form of the element, known as an isotope, changes into a stable one by ejecting a part of its nucleus.
Carbon is a useful element for dating objects because it’s so prevalent in our environment. The unstable isotope of carbon is 14C; its stable, unchanging isotope is 12C, where the numbers refer to different atomic weights. As 14C decays, the ratio of 14C to 12C in a sample changes over time. This change allows us to measure age.
We measure the rate of radioactive decay with what’s called a half-life. 14C’s half life is 5,730 years. This means that every 5,730 years, there’s half as much 14C as there was in the previous 5,730-year period. To extend this concept, in 11,460 years, there’s one-fourth the amount of 14C as there was originally.
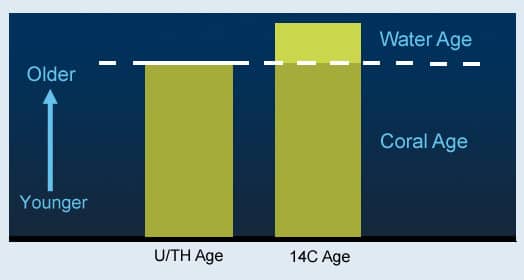
Ordinarily, we measure the age of an object by comparing the current 14C/12C ratio with ratio of the past atmosphere, since that is where radioactive carbon comes from. The difference between the two is the age since it was formed. But with deep-sea corals, that difference is both the age since the coral was formed and the age of the water in which it grew.
Since we want to know both of these values, we face the classic problem of having one measurement and two unknowns. In such cases, we need to somehow determine one of those unknowns from another angle. In the case of the deep-sea corals, we get their age by analyzing another element they contain: uranium.
Like carbon, uranium is radioactive. As it decays, however, it changes into another element, thorium. Fortunately, while a coral is growing it incorporates a lot of uranium, but no thorium. This means that as it ages its thorium/uranium ratio increases at a known rate. So, measurements of the thorium/uranium ratio provide a measurement of the coral’s age.
Now we know two things: time since the coral was formed (from uranium), and the sum of that time and the past water mass’s age (from carbon). So, the difference between these two gives us the radiocarbon age of the water.
Collecting fossil corals at different depths is like collecting water profiles today. The coral records in its skeleton all that we need to know. We just have to find the ways to tease the information out.
See Also
Dead Corals Do Tell Tales
From Oceanus magazine—Growing a little each day, coral skeletons keep a daily archive of past ocean temperatures
New Coral Dating Method Hints at Possible Future Sea-Level Changes
WHOI News Release
What Other Tales Can Coral Skeletons Tell?
From Oceanus magazine—Scientists strive to get into the genes of fossil corals to extract their history
Articles Related to Dating Corals, Knowing the Ocean
From Oceanus Magazine
From ruin to reef
As the ocean warms, a science writer looks for coral solutions
How an MIT-WHOI student used Google Earth to uncover a river–coral reef connection
Reef RX
Five marine animals that call shipwrecks home
A gift for ocean research
Counting on Corals
A cascade of life
5 unlikely ocean friendships
Can Sound Help Save Coral Reefs?
An aquatic outbreak
4 Potential Solutions for Corals in Crisis
Hope for Corals in Crisis
Reef architects
News Releases
WHOI scientist joins global Tara Coral expedition to unlock secrets of climate-resilient reefs
Seawater microbes offer new, non-invasive way to detect coral disease, WHOI-led study finds
Ship-mounted camera systems increase protections for marine mammals
New funding will boost vital reef restoration work
Sonic Youth: Healthy Reef Sounds Increase Coral Settlement
Scientists Discover Additional Healthy Deep-sea Coral Reefs and New Seamounts in the Galápagos
Innovative Techniques Provide New Means to Monitor Coral Reef Health
News & Insights
Valentine’s Day Courtship Tips from the Ocean
Recognizing Massachusetts Right Whale Day
What happens to natural gas in the ocean?
WHOI working to address ocean acidification; protect region’s vital shellfish industry
Unicorns of the Arctic face a new potential threat
For Mark Baumgartner, Whale Safe is the natural evolution of WHOI’s work with passive acoustics
Listening to fish with passive acoustics
Deciphering the Impacts of a Changing Ocean on Scallop Fisheries
Specialized camera system gives unprecedented view of ocean life
Jellyfish larger than blue whales?
Teaming up for right whales
WHOI in the News
eDNA methods give a real-time look at coral reef health
Harnessing the power of sound for coral reef restoration
Even ‘Twilight Zone’ Coral Reefs Aren’t Safe from Bleaching
UVI and UTA scientists uncover new insights on stony coral disease affecting U.S. Virgin Islands and Puerto Rico
See the Pristine Coral Reefs Found off the Galápagos Islands
While One Iconic Kelp Forest Faces An Incredible Decline, Another Offers A Glimmer Of Hope
The Mystery of the Healthy Coral Reef
Ocean Life Features

A healthy reef protects coastlines from wave damage, plays a critical role in providing food, boosts the economy, and provides…

Stressors can affect organisms living on the reef or they can affect the corals, themselves. When corals die, other organisms…

Fish that inhabit a coral reef play essential roles in the reef ecosystem, and reefs without fish struggle to recover…

Many people think of coral as hard, rock-like formations that attract abundant, diverse marine life. In fact, corals are tiny…

When most people think of corals, they think of the Great Barrier Reef off Australia, but deep in the ocean…

Coral is a useful tool for scientists who want to understand changes in past climate, but recalling that history presents…