Introduction
Ocean acidification is a new field of research in which most studies have been published in the past 10 years. Hence, there are some certainties, but many questions remain. Ocean acidification is also a multi-disciplinary research area that encompasses topics such as chemistry, paleontology, biology, ecology, biogeochemistry, modeling, and social sciences. Furthermore, some aspects of ocean acidification research, for example the carbonate chemistry, are intricate and counterintuitive. For these reasons, the media and the general public find some scientific issues or results confusing.
The U.S. Ocean Carbon and Biogeochemistry (OCB) program, supported by the European Project on Ocean Acidification (EPOCA) and the UK Ocean Acidification Research Programme, has compiled a list of frequently asked questions (FAQs). These questions were widely distributed to the research community with the request to draft concise replies summarizing current knowledge, yet avoiding jargon. The replies were then subject to an open peer-review and revision process to ensure readability without any loss of scientific accuracy. The response of the community was enthusiastic. In total, 27 scientists from 19 institutions and 5 countries contributed to the whole process.
We do hope that this FAQ list will prove useful and would like to point out that it is an on-going process. Anyone is invited to seek clarification or send comments to Sarah Cooley. The list will be revised periodically using this input.
Joan Kleypas and Richard Feely (OCB), Jean-Pierre Gattuso (EPOCA), and Carol Turley (UK Ocean Acidification Research Programme)
Related Files
» PDF of OA FAQs (364KB) -- English
» PDF of OA FAQs -- French/Francais
» PDF of OA FAQs -- Chinese/在中國
» PDF of OA FAQs -- German/Deutsch
The name "ocean acidification"
The ocean is not acidic, and model projections say the oceans won?t ever become acidic. So why call it ocean acidification?
Ocean acidification refers to the process of lowering the oceans’ pH (that is, increasing the concentration of hydrogen ions) by dissolving additional carbon dioxide in seawater from the atmosphere. The word “acidification” refers to lowering pH from any starting point to any end point on the pH scale. This term is used in many other scientific areas (including medicine and food science) to refer to the addition of an acid to a solution, regardless of the solution's pH value. For example, even though seawater's pH is greater than 7.0 (and therefore considered “basic” in terms of the pH scale), increasing atmospheric CO2 levels are still raising the ocean's acidity and lowering its pH. In comparison, this language is similar to the words we use when we talk about temperature. If the air temperature moves from -40°C to -29°C (-40°F to -20°F), it is still cold, but we call it “warming.” — James Orr, Senior Scientist, Laboratory for the Sciences of Climate and Environment, France; Christopher L. Sabine, Supervisory Oceanographer, NOAA Pacific Marine Environmental Laboratory, USA; Robert Key, Research Oceanographer, Princeton University, USA
Would dissolving all the CO2 released by burning all the world?s fossil fuel reserves ever make the seas acidic?
No. The fundamental chemistry of the ocean carbon system, including the presence of calcium carbonate minerals on the ocean floor that can slowly dissolve and help neutralize some of the CO2, prevents the oceans from becoming acidic on a global scale. — Christopher L. Sabine, Supervisory Oceanographer, NOAA Pacific Marine Environmental Laboratory, USA
Is ocean acidification just another name for climate change?
No. While ocean acidification and climate change share a common cause (increases in CO2 in the atmosphere), climate change encompasses the effects associated with changes in the Earth’s heat budget (due to the greenhouse effect of CO2 and to a lesser extent other climate reactive gases), which cause global warming and changes in weather patterns. Ocean acidification specifically refers to the lowering of ocean pH resulting from its absorption of human-released CO2 from the atmosphere. Ocean acidification does not include the warming of the ocean. — Christopher L. Sabine, Supervisory Oceanographer, NOAA Pacific Marine Environmental Laboratory, USA
Ocean carbon chemistry and pH
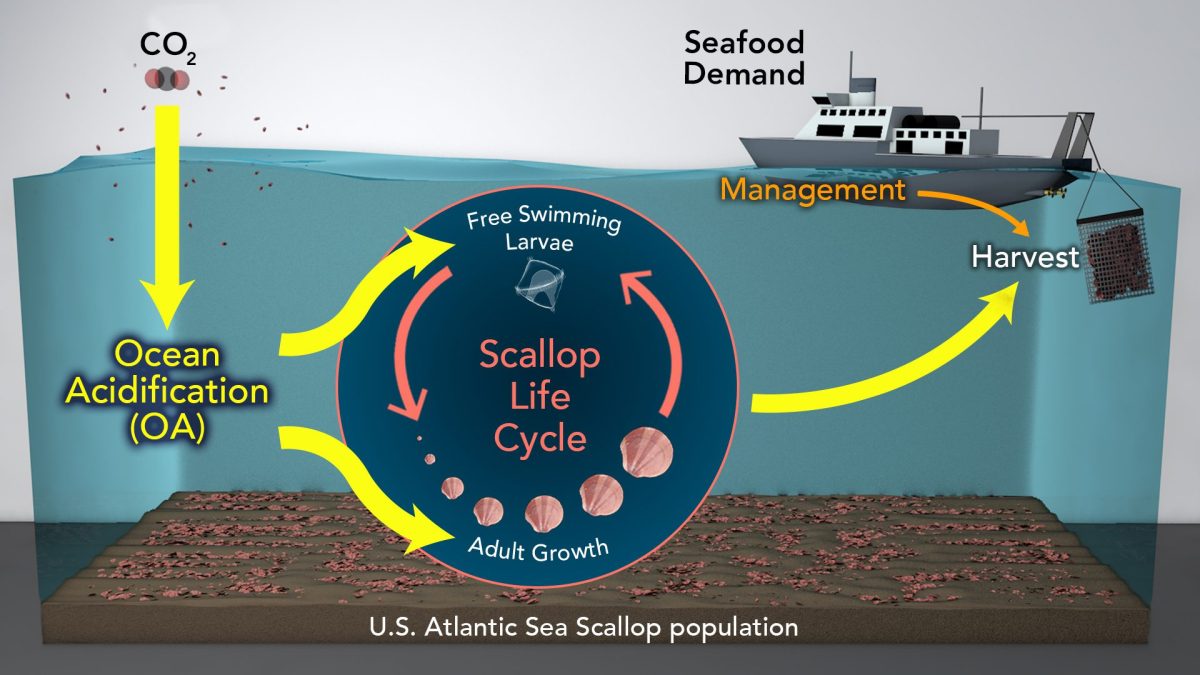
The equations showing CO2 reacting with water look like they generate more, not less carbonate. How does ocean acidification decrease the amount of carbonate ions in seawater?
This is a common point of confusion, because step-by-step equilibrium equations describing the carbonate system in seawater do not capture the dynamic chemical environment of seawater. There are several reactions that can occur between carbon dioxide (CO2), water (H2O), carbonic acid (H2CO3), bicarbonate ion (HCO3-), and carbonate ion (CO32-). One of the possible reactions does create carbonate ions and lowers pH:
CO2 + H2O ↔ H2CO3 ↔ H+ + HCO3- ↔ 2H+ + CO32-
However, at the current ocean pH level, another reaction also occurs that consumes carbonate ions and does not change pH:
CO2 + H2O + CO32- ↔ 2HCO3-
The second equation describes the reaction that occurs most often in the modern oceans, but the first reaction also occurs, so the resulting overall change is a decrease in carbonate and a decrease in pH. — Christopher L. Sabine, Supervisory Oceanographer, NOAA Pacific Marine Environmental Laboratory, USA
Will CO2 really decrease ocean pH all that much?
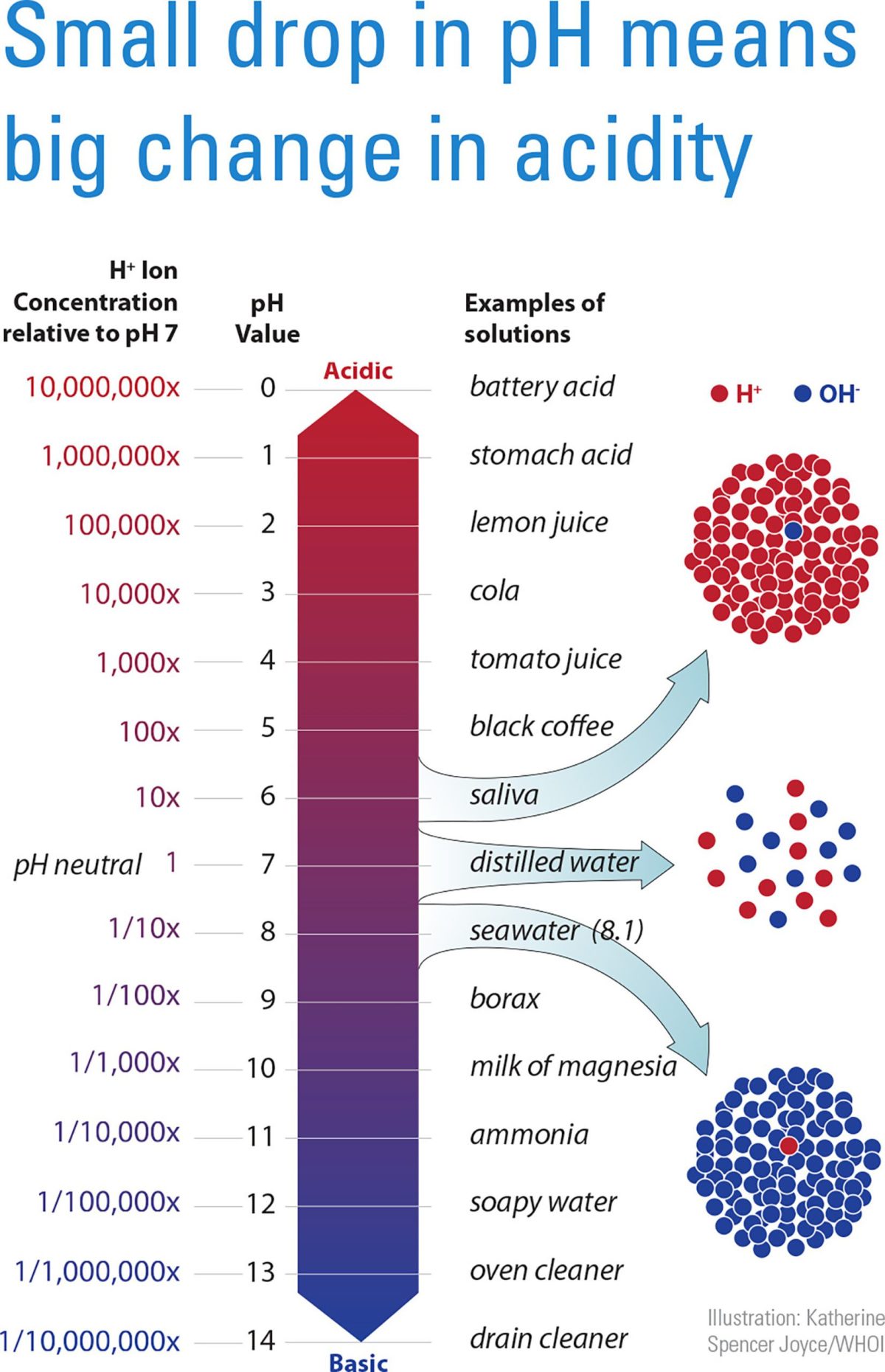
Scientists estimate that surface ocean pH has fallen by about 0.1 pH unit from preindustrial times to today. Because pH is a measure of hydrogen ion concentration and the the pH scale is logarithmic — for every drop of 1 pH unit, hydrogen ion levels increase by a factor of 10 — a 0.1-unit pH drop is equivalent to about a 26% increase in the ocean hydrogen ion concentration. If we continue on the expected trajectory for fossil-fuel use and rising atmospheric CO2, pH is likely to drop by 0.3-0.4 units by the end of the 21st century and increase ocean hydrogen ion concentration (or acidity) by 100-150% above what it was in preindustrial times. — Scott Doney, Senior Scientist, Woods Hole Oceanographic Institution, USA
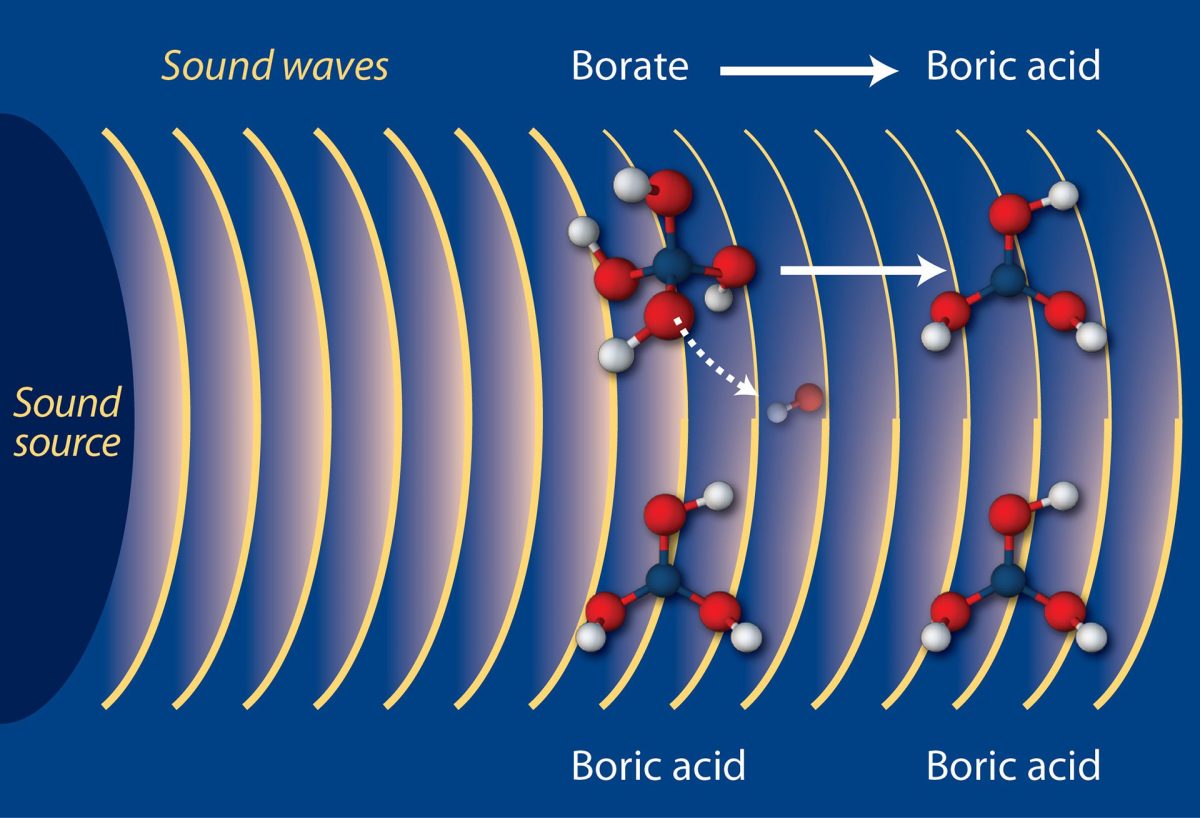
It seems impossible to acidify the oceans, given how salty they are. How could CO2 overcome all that salt?
When acids and bases neutralize each other in a laboratory experiment, salt and water form. But in the ocean, the major ions that make seawater “salty” (like sodium, chloride, and magnesium) have come from rock weathering, which provides a balanced amount of positive and negative ions to the seas over many millennia. Variations in ocean pH on shorter time scales of decades to centuries are controlled by weak acids and bases, like bicarbonate or borate. Of these weak acids and bases, the dissolved forms of CO2, known as carbonic acid, bicarbonate, and carbonate, have the largest impact on global ocean pH variations because their concentrations are changing quickly relative to other ions in the ocean. — Christopher L. Sabine, Supervisory Oceanographer, NOAA Pacific Marine Environmental Laboratory, USA
If the ice caps melt and freshwater is added to the ocean, won?t this simply dilute the acidity?
Fresh water from melting ice caps dilutes the concentrations of all the various components of the carbonate system in seawater (described above), as well as the total alkalinity and salinity (both of which affect pH). For example, a liter of “typical” Arctic seawater (temperature, 5°C; salinity, 35; total alkalinity, 2244 micromoles/kilogram) that is exposed to today’s atmospheric CO2 level of 390 ppm has a total carbon content of 2100 micromoles/kilogram and a pH of 8.04 (total scale, here and below). Adding a kilogram of freshwater to the kilogram of seawater would dilute the salinity, alkalinity, and carbon content to half of what they were, and the initial pH would increase to 8.21. However, that seawater is out of equilibrium with the atmosphere (it now has a pCO2 of 151 ppm, while the pCO2 level of the overlying atmosphere is 390 ppm) and so it will absorb CO2 until the seawater pCO2 also equals 390 ppm, at which point the pH will have dropped to 7.83.— Richard A. Feely, Senior Scientist, NOAA Pacific Marine Environmental Laboratory, USA; Joan Kleypas, Scientist III, National Center for Atmospheric Research, USA
Won't the CO2 outgas as the oceans begin to warm up, therefore cancelling out the problem?
The CO2 content of the surface waters of the oceans responds to both changes in CO2 content of the atmosphere and changes in temperature. For example, if ocean temperatures were not changing, a doubling of preindustrial CO2 levels (from 280 to 560 ppm) would cause an increase in the total amount of dissolved carbon in the surface ocean from about 2002 to 2131 micromoles/kg of seawater (assuming salinity = 35, temperature =15°C, and alkalinity = 2300 micromoles/kg). If ocean temperatures warmed by 2°C over that period, then less carbon would be taken up (the increase would be from 2002 to 2117 micromoles/kg). Thus, a 2°C increase in temperature results in about a 10% decrease in carbon uptake in surface waters. The expected warming of the oceans also may alter ocean circulation, further reducing their capacity to absorb CO2 from the atmosphere, but the excess CO2 will still remain in the atmosphere and drive further acidification. For pH, the net effects of climate warming on atmospheric CO2, CO2 solubility, and chemical speciation approximately cancel out. — Scott Doney, Senior Scientist, Woods Hole Oceanographic Institution, USA; Joan Kleypas, Scientist III, National Center for Atmospheric Research, USA
Measurements and observations
How do we know what ocean pH was in the past even though the pH scale was not introduced until 1909?
When ice sheets build up into glaciers, air bubbles become trapped in the freezing ice. Scientists have analyzed the CO2 concentration of air in these bubbles and have developed a record of the atmospheric CO2 concentration in the recent past. Because large parts of the surface ocean CO2 concentration remains roughly in equilibrium with the atmospheric CO2 concentration, the ocean CO2 content can be calculated from these air bubbles, and ocean pH can also be calculated. In fact, the ice core record shows that the atmospheric CO2 concentration has never been higher than about 280 ppm during the last 800,000 years, creating conditions leading to an average preindustrial surface ocean pH of ca. 8.2. — Jelle Bijma, Biogeochemist, Alfred Wegener Institute for Polar and Marine Research, Germany
What evidence suggests that ocean acidification is happening and that it results from human activity?
Scientists have collected semi-continuous records of seawater pCO2 and pH over the last 20-30 years in the Pacific and Atlantic Oceans. These time-series records from near Hawaii, Bermuda, and the Canary Islands show that seawater pCO2 is mirroring the increase in atmospheric CO2 and that ocean pH is decreasing. Other measurements of the CO2 content in the North Pacific Ocean, conducted in 1991 and again in 2006, show that the CO2 content in the North Pacific Ocean has increased in accordance with rising atmospheric CO2 concentrations. — Carol Turley, Senior Scientist, Plymouth Marine Laboratory, UK; Joan Kleypas, Scientist III, National Center for Atmospheric Research, USA
How do we know what ocean pH was tens of millions of years ago?
To estimate physical or chemical parameters such as temperature or pH for periods before instruments were available, scientists use so-called proxy parameters or “proxies,” which are measurable parameters that can be related to desired but unobservable parameters. For instance, marine calcifying organisms incorporate many other elements into their hard shells and skeletons besides the calcium, carbon, and oxygen in calcium carbonate. When the hard parts of these organisms that are preserved in sediment are analyzed, the additional elements provide information about environmental conditions during the animal's lifetime. Historical ocean pH values and changes can be studied using the concentration of the element boron and the ratio of its stable isotopes (δ 10B and δ 11B) in marine carbonates. Additional geochemical evidence and modeling provide strong evidence that the average surface ocean pH has not been much lower than about 8.2 for millions of years. — Jelle Bijma, Biogeochemist, Alfred Wegener Institute for Polar and Marine Research, Germany
How do OA's effects relate to those of other human activities?
Other human activities certainly are affecting seawater chemistry and the ocean’s acid-base balance, but not nearly to the extent of atmospheric CO2-driven acidification. Acid rain, which contains sulfuric and nitric acids originally derived from fossil fuel combustion, falls on the coastal oceans. The impact of acid rain on surface ocean chemistry may be important locally and regionally, but it is small globally and its total effects equal only a few percent of the changes driven by rising atmospheric CO2. Coastal waters are also affected by excess nutrient inputs, mostly nitrogen, from agriculture, fertilizers and sewage. The resulting chemical changes lead to large plankton blooms, and when these blooms collapse and sink below the surface layer the resulting respiration from bacteria leads to a drawdown in seawater oxygen and an increase in CO2, which decreases pH even more in subsurface coastal waters.
One of the major differences between OA and these types of human effects is that OA’s influence is truly global in scale, affecting pH-sensitive and calcifying organisms in every ocean basin from the equator to the poles. At present, the effects are restricted primarily to the upper 200-500m of the ocean, but every year the effects penetrate to deeper depths. Many of the other impacts of human activities are more local in nature.— Scott Doney, Senior Scientist, Woods Hole Oceanographic Institution, USA; Chris Langdon, Associate Professor, University of Miami
Geological buffering
If glacial runoff increases and rock flour is carried to the oceans, will this provide alkalinity to the oceans and offset OA somewhat?
The weathering of continental rocks does increase the alkalinity of seawater and increases its ability to counteract pH decreases, but neutralizing all of the CO2 from human activity that is entering the oceans with this process alone would take hundreds of thousands of years. Therefore, on the time scales of importance to humankind (decades to centuries), these processes are not fast enough to significantly buffer ocean acidification. — Richard A. Feely, Senior Scientist, NOAA Pacific Marine Environmental Laboratory, USA; Jelle Bijma, Biogeochemist, Alfred Wegener Institute for Polar and Marine Research, Germany
As the oceans become more acidic, more calcium carbonate minerals underwater will dissolve. Will that offset ocean acidification?
The dissolution of calcium carbonate minerals in the water column and in the sediments does increase the alkalinity of seawater, which offsets the decreased pH and carbonate ion concentrations associated with ocean acidification. However, as with rock weathering, this process is slow and would take thousands to tens of thousands of years to neutralize all of the CO2 from human activity that is entering the oceans. Over the decades to centuries that affect human communities, these processes are not fast enough to counteract CO2 invasion into the ocean, and so the chemical changes associated with ocean acidification will last for several centuries. — Richard A. Feely, Senior Scientist, NOAA Pacific Marine Environmental Laboratory, USA
OA and photosynthesis
Photosynthesis is expected to rise with ocean CO2 levels, and corals contain photosynthesizing algae, so won't corals benefit from rising CO2?
The photosynthesis of some, but not all, algae increases when CO2 rises to levels projected for the end of this century (700-800 ppm). The single-celled algae called zooxanthellae that live within coral animals’ cells are some of the algae whose photosynthesis does not significantly increase at projected future CO2 levels. Normally, zooxanthellae and corals maintain a delicately balanced symbiosis, in which the zooxanthellae transfer photosynthetically formed carbon-based nutrition to the coral host and provide an important source of carbon for the coral and for coral calcification (skeleton building). If the algae within the corals’ cells do too well and their numbers greatly increase, the transfer of nutrition to the coral host can be disrupted. So even if zooxanthellae photosynthesis were to increase under high CO2, this does not necessarily benefit the corals. In the great majority of experiments, coral calcification rate decreases when the CO2 level increases, so it is clear that the rise in CO2 is decreasing the corals’ ability to build their skeletons rather than protecting them by altering zooxanthellae photosynthesis. — Chris Langdon, Associate Professor, University of Miami, USA; Anne Cohen, Research Specialist, Woods Hole Oceanographic Institution, USA
If photosynthesis increases with ocean CO2 levels, won?t phytoplankton and seagrasses do better?
Communities of organisms found near shallow near-shore volcanic CO2 vents demonstrate that certain microalgae, seaweeds and seagrasses grow very well in areas that experience long-term exposure to elevated CO2. However, this work also shows that coastal ecosystems are degraded due to the long-term effects of ocean acidification. Biodiversity is lost: groups of organisms such as coralline algae gradually disappear as pH falls, and they are replaced by thriving stands of invasive algae. This raises concerns that ocean acidification will allow alien algae to proliferate and disrupt coastal habitats. — Jason Hall-Spencer, Lecturer, University of Plymouth, UK
An increase of CO2 in seawater increases growth of photosynthetic algae? Isn't that a good thing?
The growth and photosynthesis of certain marine phytoplankton and plant species may increase with higher CO2 levels, but this is by no means a general rule. For other species, higher CO2 and rising acidity will have either negative or neutral effects on their physiology. Therefore some marine phytoplankton and plants will be “winners,” while others will be “losers.” This means that instead of benefiting all impartially, future acidification will instead probably cause major shifts in the species composition of ocean phytoplankton communities. Some of the experiments that have been done so far suggest that the likely new dominant phytoplankton species in the future acidified ocean may be less able to support the productive food chains that we presently rely on to support healthy ocean ecosystems and fisheries resources. — David Hutchins, Professor of Marine Environmental Biology, University of Southern California, USA
OA and calcification
Why does adding CO2 to home aquaria benefit animals, but in the ocean, adding CO2 leads to harmful acidification?
Freshwater fish and plants tend to be more tolerant of lower pH and wider pH changes overall because fresh water contains low alkalinity, which means that the water chemistry does not minimize pH changes (i.e., it does not have the “buffering capacity”) the way that seawater chemistry does. The natural variability of pH in lakes and rivers is also higher than in the ocean. Freshwater organisms have evolved special mechanisms that allow them to thrive in these more acidic and variable conditions; for example, freshwater plants may benefit from higher CO2.
In saltwater aquaria, corals and fish require a more narrowly balanced pH and owners often add carbonate “hardeners” to increase the water's alkalinity and maintain the pH between 8.0 and 8.4. Devices called “calcium reactors” bubble CO2 gas through crushed calcium carbonate (usually crushed coral), which releases calcium and carbonate ions into the salt water, providing the high-alkalinity, calcium-rich waters that aquarium corals and other calcifying organisms need to continue healthy growth. Unfortunately, these types of devices cannot be used to solve ocean acidification on a global scale, because of the vast amounts of crushed calcium carbonate that are required to carry out the process in the world’s oceans. — Helen Findlay, Lord Kingsland Fellow, Plymouth Marine Laboratory, UK; Joan Kleypas, Scientist III, National Center for Atmospheric Research, USA; Michael Holcomb, Postdoctoral Research Associate, Centre Scientifique de Monaco, Monaco
Shellfish can survive in fresh water where pH can drop as low as 5, so what?s the problem?
Organisms that live in fresh water or in salt water with lower pH have developed adaptive mechanisms that allow them to survive under those conditions. In contrast, marine shellfish that have evolved in seawater with a higher and less variable pH are more susceptible to changes in pH. A good example of this is the natural shift in marine organisms to freshwater organisms living along estuaries. A marine shellfish, Thais gradata, that is found along estuaries tends to have higher rates of dissolution at the freshwater end of the estuary, where pH is lower and varies widely, than it does at the seawater end of the estuary where pH is higher and varies less. — Helen Findlay, Lord Kingsland Fellow, Plymouth Marine Laboratory, UK
Why does increasing the dissolved CO2 concentration in seawater affect shell building in marine organisms?
Dissolving CO2 in seawater causes a suite of changes in the carbonate system in seawater: the concentrations of dissolved CO2, total dissolved inorganic carbon, and the bicarbonate ion increase, while pH, carbonate ion concentration, and calcium carbonate saturation state decrease. One or several of these changes may affect shell building in marine organisms. The formation of skeletons or shells in most marine organisms is an internal process where most organisms appear to convert bicarbonate to carbonate to form calcium carbonate. But because this conversion creates protons (hydrogen ions), the organisms must exert energy to expel the hydrogen ions into the external environment (seawater). One hypothesis as to why ocean acidification can cause slower calcification rates (and there are several) is that as seawater pH decreases, the organisms must exert more energy to rid themselves of the protons produced by calcification — they are simply working against a steeper gradient. This explains why many calcifying organisms have lower calcification rates when they are physiologically taxed by other stresses (e.g. lack of food); that is, the added stress leaves the organisms with less energy for calcification. Ocean acidification can also indirectly affect shell formation through physiological impacts, such as changes in an organism's respiration rate, which can impact energy budgets and thus alter the animal's ability to produce shell material. While some organisms may grow their shells at normal rates under ocean acidification, the exposed parts of the shell may dissolve more quickly, so that the organism may need to spend more energy in shell maintenance, and less in reproduction or other life activities. — Helen Findlay, Lord Kingsland Fellow, Plymouth Marine Laboratory, UK; Anne Cohen, Research Specialist, Woods Hole Oceanographic Institution, USA; Joan Kleypas, Scientist III, National Center for Atmospheric Research, USA
Scientists have shown that lobster shells (and shells of other food-providing animals) get thicker when living in water with higher CO2, so why should we be worried about OA?
At least one experimental study showed that the shell mass of several crustaceans, including lobsters, reared in culture for 60 days actually increased with increased CO2. Shell-making requires energy, so increased shell mass almost certainly occurred hand-in-hand with reduced energy for other functions like growth and reproduction. Also, lobsters and other crustaceans make shells using both calcium carbonate and chitin in a different mechanism than other marine organisms. They shed their shell periodically, rather than growing constantly throughout life, and they are thought to retain many of the minerals from their old skeleton to put into the new skeleton. Energy and mineral budgets were not monitored in the above study, so how OA affects the overall health and longevity of these organisms is still not known. — Anne Cohen, Research Specialist, Woods Hole Oceanographic Institution, USA; Helen Findlay, Lord Kingsland Fellow, Plymouth Marine Laboratory, UK
Individuals and Ecosystems
Won't organisms and ecosystems adapt if some calcifying species leave an area?
The rate of human-driven ocean acidification is about 100 times faster in the surface ocean than that experienced by marine ecosystems globally for tens of millions of years. Different ecosystems will respond differently. In some ecosystems, such as coral reefs, the calcifying organisms form the fundamental architecture of the ecosystem so if they disappear, the ecosystem could disappear. In other ecosystems where calcifiers play a less important role, it is less clear what impact the loss of calcifying species might have on the ecosystem. During profound rapid changes in ocean chemistry like present-day ocean acidification, organisms respond in one of 3 ways: acclimation, adaptation, or extinction. If most species acclimate rapidly, the biodiversity and function of marine ecosystems may be relatively unchanged. Evolutionary adaptation, however, is linked to generation time, meaning that long-lived species that mature slowly will have fewer opportunities to produce offspring more resistant to the rapidly changing environmental conditions. Even species that reproduce more quickly may not be able to adapt; for example, at the edges of regions with favorable temperatures and water chemistry, corals have been trying to adapt to lower carbonate ion concentrations for many millions of years, but they have not been able to succeed in outcompeting algae and other non-calcifying species there. It seems unlikely, therefore, that corals could succeed in adapting to new temperatures and water chemistry in a few decades to respond to OA. If OA drives large shifts in the abundance of key organisms in food webs, or significant rates of extinction, we can expect important changes in the function of ecosystems-- how energy and material flow from primary producers like plankton to top predators like fish and mammals.
Ecosystems are complex networks of interactions among biological organisms and the environment, and it is difficult to predict the full ecological impacts of changing any of those links. We know from CO2 vent studies that OA affects biological species differently and the mix of marine species shifts, leading to lowered biodiversity and a change in the overall functioning of ecosystems. We depend on a whole range of marine ecosystem services, including food from fisheries, income from tourism and recreation, and oxygen and nutrient recycling from biogeochemical processes; all of these services could be altered, and in many cases degraded, by ocean acidification. Imagine, for example, the economic effects of the disappearance of sea urchins from Japanese fisheries or the decline in fish larvae of commercially important species. Furthermore, decreasing or disappearing calcifying organisms will affect (1) the chemical environment, (2) other calcifying and non-calcifying organisms that may depend on them (e.g., many organisms and hundreds of millions of people depend on coral reefs), and (3) the reservoir of carbon on Earth (the “rock” produced by calcifying organisms falls on the ocean floor to form massive “chalky” deposits that lock away some carbon into geological structures). Just like a neglected aquarium that gives way from fish and shellfish to algae, marine ecosystems may adjust, but they might then be populated by species that are less useful or desirable to humans, making the traditional resources and services provided by the changed ecosystems unavailable, different from before, or unpredictable.— Debora Iglesias-Rodriguez, Lecturer, National Oceanography Centre of the University of Southampton, U.K; Scott Doney, Senior Scientist, Woods Hole Oceanographic Institution, USA; Steve Widdicombe, Benthic Ecologist, Plymouth Marine Laboratory, UK; Jim Barry, Senior Scientist. Monterey Bay Aquarium Research Institute, USA; Ken Caldeira, Senior Scientist, Carnegie Institution for Science, USA; Jason Hall-Spencer, Marine Biology Lecturer, University of Plymouth, UK
Will ocean acidification kill all ocean life?
No. However, many scientists think that ocean acidification will lead to important changes in marine ecosystems. This prediction is largely based on geologic history: millions of years ago, marine ecosystems experienced rapid changes during ocean acidification events, including some species extinctions (see “OA in Geologic History” below). Today, some species and the ecosystems they sustain are threatened by ocean acidification, particularly in combination with other climate changes such as ocean warming. Examples include tropical corals, deep-sea corals, and swimming snails. These species play key roles in the oceans either because they build three-dimensional structures, which host a considerable biodiversity, or because they are key components of the food chain. Some species that build calcium carbonate structures, such as coral reefs, also provide key services to humans by providing food, protecting shorelines, and supporting tourism. Evidence for the ecological effects of ocean acidification today can be found at “champagne sites,” locations where volcanic CO2 vents naturally acidify the water and small CO2 bubbles rise through the water column. At one of these sites around the Island of Ischia (Italy), for example, biodiversity is reduced by 30% at the acidity level that matches the level expected globally in 2100. — Jean-Pierre Gattuso, Senior Scientist, Centre National de la Recherche Scientifique and Université Pierre et Marie Curie-Paris 6, France; Jason Hall-Spencer, Marine Biology Lecturer, University of Plymouth, UK
Will warming and acidification balance out responses from organisms?
In principle, there may be some benefit from warming for the calcification process, because precipitation of calcium carbonate is enhanced by temperature up to a certain threshold. However, organisms are accustomed to living in a limited thermal range and are performing less well in temperatures outside of this range. In many marine areas, organisms (calcifiers and non-calcifiers alike) are already exposed to temperatures reaching the upper end of their thermal windows. Pilot studies on crab and fish have demonstrated that exposure to CO2 levels expected if CO2 emissions continue to increase reduces the animals’ capacity to tolerate extreme temperatures. Studies on corals have also shown that CO2 enhances thermal sensitivity. In this case it encourages the likelihood of bleaching events triggered by warming. Overall, it appears that ocean acidification may enhance the sensitivity of organisms to climate warming. — Hans-Otto Pörtner, Professor, Alfred Wegener Institute, Germany
Will adult organisms be safe if they are able to survive the effects of ocean acidification when they are very young and susceptible?
For common marine organisms, the gametes, eggs, various larval stages, juveniles, and adults may be affected differently by ocean acidification because they have different tolerances and coping strategies to environmental stress. In some cases, the early life stages may be more susceptible to stress, while in other cases, the adults may be. Experiments are necessary on all life stages to understand the full effects on an organism and to highlight stages that represent weak links. It is also important to consider ocean acidification's lifelong impacts on survival and reproduction. In general, early life history phases (gametes, larvae, juveniles) are expected to be more sensitive to ocean acidification than adults. If fewer young organisms survive to adulthood, population size will clearly be reduced. Ongoing stress usually limits the success of organisms – for example, stressed organisms grow slower and smaller, stressed predators will be less effective, and stressed prey may be less able to avoid capture – and ultimately this stress will decrease survival, causing population size to suffer. For adults, stress caused by ocean acidification may not affect everyday activities, but it will ultimately reduce organisms' growth and reproduction rates. Decreased reproduction can also alter the entire population's size. Impacts at any life stage can reduce the potential for a population to grow or to recover from losses due to disturbance or stress. — Jim Barry, Senior Scientist. Monterey Bay Aquarium Research Institute, USA; Helen Findlay, Lord Kingsland Fellow, Plymouth Marine Laboratory, UK
CO2 is a normal product of respiration. Animals breathe it in and out all the time. How can it possibly be toxic?
Just as in seawater, respiratory CO2 reduces the pH within cells. Organisms have evolved mechanisms to buffer, transport, and remove CO2 from their cells at the rate at which it is produced. Ocean acidification reduces the CO2 difference between the inside and outside an animal's body, thereby hindering CO2 removal and causing “respiratory acidosis.” (This term is analogous to “ocean acidification” because normal bodily fluids are slightly basic.) Respiratory acidosis may lead to, among other things, reduced metabolism and reduced organism activity. Additionally, many cellular functions are pH sensitive and may respond negatively to respiratory acidosis. For example, respiratory proteins (e.g. hemoglobin) in the blood bind oxygen at high pH and release it at low pH, allowing oxygen uptake at the gills and release at the cells, where metabolically produced CO2 has decreased local pH. Many organisms can compensate for respiratory acidosis by shifting the balance of ions in the body. However, it is unknown whether they can maintain such an ionic imbalance in the long term. — Brad Seibel, Assistant Professor of Biological Sciences, University of Rhode Island, USA
OA in geologic history
Why would coral become extinct because of ocean acidification, when coral species have already survived other ocean chemistry changes over geological history?
The danger from ocean acidification is related to the current rate of change, the concentration of atmospheric CO2 expected, and the magnitude of change of atmospheric CO2 forecast if we keep emitting CO2 at the same rate. The present rise in atmospheric CO2 is ~2 ppm per year, and atmospheric CO2 has increased more than 100 ppm since the beginning of the Industrial Revolution. In the transition between the end of the last ice age to the current warm period, CO2 concentrations increased 80 ppm occurred over more than 10,000 years. Today’s rates of CO2 increase in the atmosphere are therefore approximately 100 times greater than most changes sustained over geologic time. Other than at times of the great mass extinctions, there is no evidence in the geologic record for sustained rates of change in atmospheric CO2 that have been as great or greater than today’s. Even during extreme ocean chemistry changes in geological history—- for example, during the Paleocene/Eocene thermal maximum 55 million years ago (Ma) when carbonate minerals dissolved in most of the deep and intermediate ocean—-these changes probably happened over several thousands of years. Corals have indeed survived multiple extinction events in Earth history, but each time their “rebound” took millions of years, and their ability to form reefs took even longer. The earliest corals arose during the Ordovician more than 400 million years ago. Known as Tabulate and Rugose corals, these were very different from the corals living today (modern corals belong to the Scleractinia and likely evolved independently from these earlier forms), and the Ordovician reef systems were dominated by sponges rather than corals. These groups went extinct during the Permo-Triassic extinction event 251 million years ago, and different coral lines eventually evolved and flourished again, along with reef-building bivalves that built tremendous reefs during through the Cretaceous period, most of which went extinct (along with the dinosaurs) in the Cretaceous extinction event 65 Ma ago. While coral reefs disappeared at this time, about half of all coral species did survive, but it took millions of years before reefs recovered to become widespread once again. In general, ocean life recovers from extinction episodes by adaptation and evolution of new species, but this takes roughly 10 million years to achieve pre-extinction levels of biodiversity. — Jim Barry, Senior Scientist, Monterey Bay Aquarium Research Institute, USA; Daniela Schmidt, Senior Research Fellow, University of Bristol, UK; Ken Caldeira, Senior Scientist, Carnegie Institution for Science, USA
How is today's change in ocean chemistry different from those of previous geological periods?
Present conditions differ from the past largely because the rate of change of atmospheric CO2 does not match the rate of mitigating geological processes. If CO2 is added slowly over hundreds of thousands of years, as it was during the Ordovician by volcanic and plate tectonic activity, the CO2 that enters the ocean has time to mix throughout the ocean from top to bottom. As a result, even though the amount of CO2 that is taken up by the ocean is large, it is spread out over a very great volume of water and the resulting decrease in pH is small. At the same time, as the CO2 level in deep oceans increases over millennia, carbonate sediments lying on the seafloor begin to dissolve and release carbonate ions that neutralize some of the acidity, further minimizing the decrease in pH. Past oceans also contained higher calcium and magnesium ion concentrations, which helped stabilize calcium carbonate minerals in marine animals’ skeletons.
Today, the CO2 in the atmosphere is increasing much faster than the ocean mixes. During CO2 releases like this over “short” (<10,000 year) timescales, the ability of sediments to regulate ocean chemistry is overwhelmed and both pH and saturation state decline. Even though the amount of CO2 that has entered the ocean in the last 200 years is smaller than that added during the Ordovician, the CO2 has built up to a much higher concentration in the surface ocean. As a result, upper ocean pH has decreased more rapidly and by a greater amount than in the geological past. Both the rate of change of pH and the magnitude of the change present problems for organisms that evolved in an ocean that experienced smaller, slower pH changes in the past. — Chris Langdon, Associate Professor, University of Miami, USA; Andy Ridgwell, Royal Society University Research Fellow, Bristol University, UK; Richard Zeebe, Associate Professor, University of Hawaii at Manoa, USA; Daniela Schmidt, Senior Research Fellow, University of Bristol, UK
Scientific methods
Experiments on organisms are often unrealistic, because scientists sometimes add mineral acids and not CO2 to lower the pH to predicted levels.
When seawater is manipulated through addition of mineral acid, and this is accompanied by the addition of equal (equimolar) amounts of sodium bicarbonate, this approach perfectly simulates the changes in seawater carbonate chemistry induced by CO2 uptake. Even when ocean acidification is simulated by manipulating seawater with mineral acids without adding bicarbonate (or carbonate), it is almost indistinguishable in terms of pH, pCO2, carbonate ion concentration, and saturation state from seawater manipulated through CO2 aeration. The different treatments do result in slightly higher bicarbonate concentrations in CO2-aerated manipulations. However, bicarbonate increases in both approaches. In fact, no systematic difference is found in the responses of calcifying organisms exposed to seawater acidified by mineral acid or through CO2 aeration. — Ulf Riebesell, Professor of Biological Oceanography, Leibniz Institute of Marine Sciences IFM-GEOMAR, Germany
Even scientists admit there are uncertainties about climate change. How certain is ocean acidification?
There is no argument that seawater chemistry is changing due to rising atmospheric CO2, and that human combustion of fossil fuels and deforestation are the root cause. There is less certainty about the possible biological impacts of ocean acidification, but this primarily reflects the fact that different groups of marine organisms express a wide range of sensitivity to changing seawater chemistry. There is broad agreement among the scientific community that ocean acidification is occurring and that it likely will have significant effects, some positive and some negative, on a large number of marine organisms. — Scott Doney, Senior Scientist, Woods Hole Oceanographic Institution, USA
The "evidence" about ocean acidification is conflicting, so even the scientists cannot agree.
There is no disagreement in the chemical data, which show that ocean acidification is happening. However, biological data show varied responses among organisms to OA. It can sometimes seem odd that experiments conducted on the same species can produce apparently contrasting results. However, it is clear that the response of marine organisms to elevated levels of CO2 is influenced not only by the organism's identity but also by the environmental conditions it has experienced during its life. Consequently, members of the same species collected from different areas, populations, or strains can exhibit different responses. This evidence should not be considered as conflicting, but rather an insight into the natural variability that exists among populations. Only by measuring and understanding this variability will we be better able to predict which species, communities, and ecosystems are at greatest risk from ocean acidification. — Steve Widdicombe, Benthic Ecologist, Plymouth Marine Laboratory, UK
Could the observed impacts of ocean acidification result from experiments that have simply placed them straight into water with CO2 levels that will take decades or centuries to reach, which amount to little more than a shock treatment?
In ocean acidification response experiments, animals are usually not placed immediately in CO2-enriched waters, but instead they are kept in water that is then equilibrated with carefully controlled gas mixtures. Although it is impossible to perform experiments that simulate the rate of anthropogenic CO2 accumulation in the atmosphere and the oceans, the CO2 levels used are far below those that have been shown to cause shock. Nonetheless, these CO2 levels may disturb physiological processes (acid-base regulation, development of larvae, growth) in ways that appear relatively mild on short time scales. Therefore, long-term exposures are usually needed to find out whether these levels are detrimental and cause fatalities. On long time scales, even small decreases in individual animals' health may harm a species, for example, in cases where species compete with others in ecosystems or when they are exposed to another stressor like extreme temperature. — Hans-Otto Pörtner, Professor, Alfred Wegener Institute, Germany
Geoengineering and mitigation
If we increase aquaculture and grow more shellfish, won't the shells help lock up carbon dioxide (like trees)?
The calcification process does take up carbon, but it causes shifts in the carbon system in seawater that result in a lower pH and an increase in CO2 rather than its removal. Many organisms convert bicarbonate to the carbonate they use to build their shells, and this produces hydrogen ions, thus increasing acidification. Most coral reefs, for example, on the time scales we are interested in, are small sources of CO2 to the atmosphere rather than sinks. From an ecosystems point of view, even well-intended aquaculture could cause unintentional harm by altering coastal landscapes, increasing pollution and disease, or releasing genetically altered or foreign species into the environment. Any activity aimed at reducing ocean acidification should be considered in a wider context to avoid replacing one environmental impact with another. — Anne Cohen, Research Specialist, Woods Hole Oceanographic Institution, USA; Steve Widdicombe, Benthic Ecologist, Plymouth Marine Laboratory, UK
Can geoengineering solutions for climate change also help OA?
Most proposed geoengineering approaches to limit climate change attempt to provide symptomatic relief from climate change without addressing the root cause of the problem — excess carbon dioxide in the environment. Most geoengineering proposals address the climate consequences of our carbon dioxide emissions but do not address the chemical consequences of these emissions. For example, strategies that seek to cool the Earth by reflecting additional sunlight to space would have little direct effect on ocean chemistry and therefore would not significantly diminish ocean acidification.
Some proposals have sought to diminish changes in ocean chemistry by adding compounds to the ocean that would chemically neutralize acids. However, reversing ocean acidification this way would require adding an amount of material much larger than the amount of carbon dioxide we are emitting to the atmosphere. Therefore, these proposed solutions would require a new mining and chemical processing infrastructure as large as our current energy system. It seems reasonable to suggest that this level of effort and spending would be better applied to transforming our energy system away from dependence on a finite pool of fossil fuel resources to use of renewable, infinite resources— which would also prevent carbon dioxide from entering the environment in the first place instead of requiring us to consider neutralizing its effects after it is already spreading through the atmosphere and oceans. — Ken Caldeira, Senior Scientist, Carnegie Institution for Science, USA
Will capping atmospheric CO2 at 350 or 400 ppm stop OA?
Atmospheric CO2 is already at 390 ppm and is increasing at about 2 ppm per year. Without dramatic reductions in CO2 emissions, atmospheric CO2 will continue to rise, and most emission forecasts for the near future indicate a likely increase (rather than decrease) in atmospheric CO2 growth rate. The first step in addressing ocean acidification, therefore, is to stabilize and eventually reduce CO2 emissions. Atmospheric CO2 almost certainly will peak well above 400 ppm, because we will not stop increasing emissions in the next 5 years. The impacts on marine life at the peak CO2 level may be substantial. In the long run, it may be possible to reduce atmospheric CO2 through natural and artificial uptake mechanisms. The chemistry of seawater is reversible, and returning to 350-400 ppm would return pH and carbonate saturation levels to approximately their current conditions. However, some research has suggested that even current-day conditions may be deleterious for some organisms, and it is even less clear if future biological impacts due to peak CO2 will be reversible. Even if we stabilized CO2 emissions, atmospheric fossil fuel CO2 will continue to penetrate into the deep ocean for the next several centuries, which may impact deep water organisms such as cold-water corals. — Scott Doney, Senior Scientist, Woods Hole Oceanographic Institution, USA
Policy development and decision-making
Isn't it better that we sacrifice the oceans and let them keep on taking up CO2 and buffering climate?
Ocean acidification and climate change are two sides of the same coin. Both are direct consequences of anthropogenic CO2 emissions and cannot be separated from each other. The present uptake of about one quarter of anthropogenic CO2 emissions by the ocean indeed serves as a buffer against rising atmospheric CO2, and so this “service” could be considered to diminish, but not prevent, climate change. In the long term, on time scales of tens of thousands of years, the majority of anthropogenic CO2 emissions (80-90%) will end up in the ocean. This, however, will not protect the climate system from global warming during the intervening period. It is also important to point out that the impacts of CO2 uptake by the oceans will have profound effects on the functioning of Earth’s ecosystems. The oceans provide vital roles in biogeochemical cycles---not only in the regulation of CO2, but in the production of oxygen, the cycling of nitrogen and other important nutrients, as well as the production of gases that affect such things as cloud formation. Many species use both land and ocean habitats, and many humans rely on healthy oceans for their livelihoods. The oceans are an integral, interconnected part of the Earth system, and cannot be realistically considered as a separate entity. — Ulf Riebesell, Professor, Leibniz Institute of Marine Sciences IFM-GEOMAR, Germany; Joan Kleypas, Scientist III, National Center for Atmospheric Research, USA
Is it too late to do anything?
It is within our technical and economic means to modify our energy and transportation systems and land-use practices to largely eliminate carbon dioxide emissions from our economies by mid-century. It is thought that the cost of doing this — perhaps 2% of the worldwide economic production — would be small, yet at present it has proven difficult for societies to decide to undertake this conversion. — Ken Caldeira, Senior Scientist, Carnegie Institution for Science, USA
Why is it important to conduct research on OA? And what can scientists do?
Compared to our terrestrial environments, the oceans and their ecosystems are poorly understood. With recent technological advances, our knowledge is rapidly growing; however, we still have much to learn. If policymakers are to make informed decisions regarding climate change and ocean acidification, scientists need to give them the best information possible. That requires research. Everyone must recognize that obtaining and distributing that knowledge takes a great deal of effort, and maintaining clear, open communication among researchers, leaders, and citizens is critical.
Scientists have answered the question “Is ocean acidification real?” --- yes. We are now confronted with the questions, “How bad will it be?” and, “What can be done?” Most scientists agree that reducing greenhouse gas emissions is the best answer to the latter. The remaining question is the most difficult yet most important question to answer, because we are keenly aware that CO2 levels will continue to rise in the foreseeable future. Many scientists are now focused on what CO2 concentration is considered “dangerous” to the planet and to society. Addressing “What can be done?” has shifted from what can be done about the cause of the problem, CO2, to what can be done about its consequences. Essentially, we seek to answer the question, “What will future marine ecosystems look like, and what ecosystem services will they provide to the planet and humankind?” This is a huge challenge. As evidenced by many of these questions, ocean acidification is a simple problem with complex consequences. — Joan Kleypas, Scientist III, National Center for Atmospheric Research, USA; Carol Turley, Senior Scientist, Plymouth Marine Laboratory, UK; Robert Key, Research Oceanographer, Princeton University, USA
If ocean acidification is so potentially serious why isn?t it included in the United Nations Framework Convention on Climate Change (UNFCCC) Conference of the Parties (COP) climate mitigation negotiations?
Although scientists have known for decades that ocean acidification would occur as CO2 increased in the atmosphere, the consequences to marine life were not realized until about 10 years ago. At that time, biologists discovered that ocean acidification affected the ability of many marine organisms to form their shells or skeletons. Since then, many more effects of ocean acidification have been found to influence a wide array of organisms and marine processes. Because the scientific process relies on formal research protocols, peer-review, and publishing, it takes some time for a new finding to be verified and accepted by the scientific community. However, sufficient evidence about ocean acidification existed by 2007 that the IPCC Fourth Assessment Report on Climate Change (2007) stated in the Summary for Policy Makers, “The progressive acidification of the oceans due to increasing atmospheric carbon dioxide is expected to have negative impacts on marine shell-forming organisms (e.g. corals) and their dependent species.” Ocean acidification and its effects have now been documented to the point that they are widely accepted by the scientific community and it will be seriously addressed by the Fifth Assessment Report of the IPCC. In fact, ocean acidification was a major topic of discussion at side events such as Oceans Day at the December 2009 COP15 climate change negotiations in Copenhagen even though specific considerations about oceans had little or no mention in the text of the proposed agreement. — Joan Kleypas, Scientist III, National Center for Atmospheric Research, USA; Carol Turley, Senior Scientist, Plymouth Marine Laboratory, UK
Contributors and reviewers for this document
Jim Barry, Senior Scientist. Monterey Bay Aquarium Research Institute, USA
Jelle Bijma , Biogeochemist, Alfred Wegener Institute for Polar and Marine Research, Germany
Ken Caldeira, Senior Scientist, Carnegie Institution for Science, USA
Anne Cohen, Research Specialist, Woods Hole Oceanographic Institution, USA
Sarah Cooley, Postdoctoral Investigator, Woods Hole Oceanographic Institution, USA
Scott Doney, Senior Scientist, Woods Hole Oceanographic Institution, USA
Richard A. Feely, Senior Scientist, NOAA Pacific Marine Environmental Laboratory, USA
Helen Findlay, Lord Kingsland Fellow, Plymouth Marine Laboratory, UK
Jean-Pierre Gattuso, Senior Scientist, Centre National de la Recherche Scientifique and Université Pierre et Marie Curie-Paris 6, France
Jason Hall-Spencer, Marine Biology Lecturer, University of Plymouth, UK
Michael Holcomb, Postdoctoral Research Associate, Centre Scientifique de Monaco, Monaco
David Hutchins, Professor of Marine Environmental Biology, University of Southern California, USA
Debora Iglesias-Rodriguez, Lecturer, National Oceanography Centre of the University of Southampton, UK
Robert Key, Research Oceanographer, Princeton University, USA
Joan Kleypas, Scientist III, National Center for Atmospheric Research, USA
Chris Langdon, Associate Professor, University of Miami, USA
Daniel McCorkle, Associate Scientist, Woods Hole Oceanographic Institution, USA
James Orr, Senior Scientist, Laboratory for the Sciences of Climate and Environment, France
Hans-Otto Pörtner, Professor, Alfred Wegener Institute, Germany
Ulf Riebesell, Professor, Leibniz Institute of Marine Sciences IFM-GEOMAR, Germany
Andy Ridgwell, Royal Society University Research Fellow, University of Bristol, UK
Christopher L. Sabine, Supervisory Oceanographer, NOAA Pacific Marine Environmental Laboratory, USA
Daniela Schmidt, Senior Research Fellow, University of Bristol, UK
Brad Seibel, Assistant Professor of Biological Sciences, University of Rhode Island, USA
Carol Turley, Senior Scientist, Plymouth Marine Laboratory, UK
Steve Widdicombe, Benthic Ecologist, Plymouth Marine Laboratory, UK
Richard Zeebe, Associate Professor, University of Hawaii at Manoa, USA