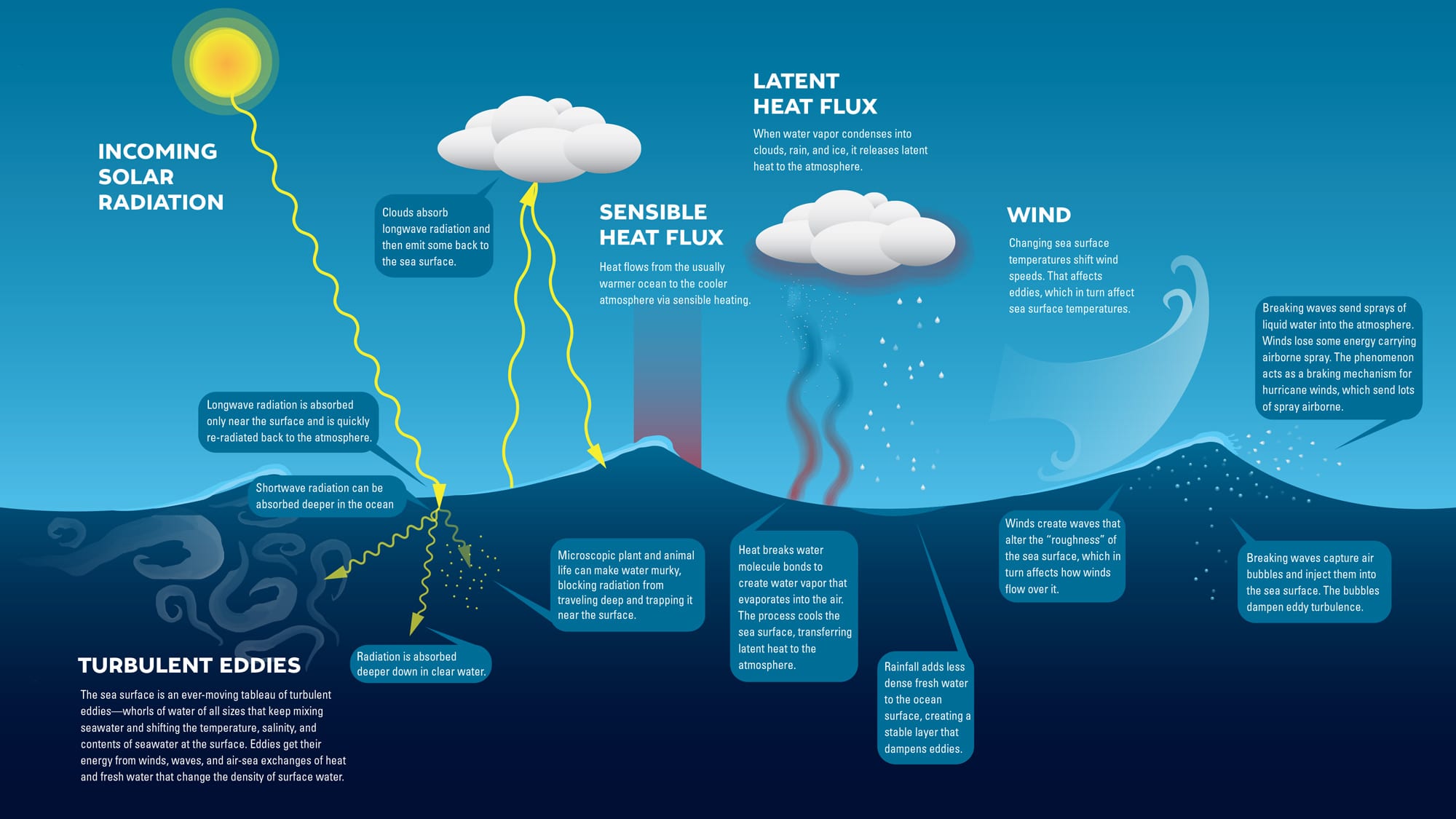
If you go swimming in a lake, the water is fresh; when you dive into the ocean, it tastes salty. But why? The water flowing into the ocean comes from freshwater streams and rivers. These bodies of water do contain salt. It dissolves from rocks on land. That’s because rain is slightly acidic. It reacts with carbon dioxide in the air to make carbonic acid. When rain falls onto rock, the acid dissolves tiny bits of salts and minerals. These wash into streams, lakes, and rivers.
But those bodies of water don’t seem salty—not like the ocean. Rain may add tiny amounts of salt, but it adds far more freshwater to streams, rivers, and lakes. When rain falls on a regular basis it keeps these waters from becoming salty.
When freshwater flows into the ocean, it carries salts and minerals with it. More salts and minerals are added from seafloor vents. Deep in the ocean, water seeps into cracks in the Earth’s crust. There, it’s heated by magma. Hot water dissolves salts and minerals from the rock. (This is similar to dissolving table salt or sugar in water. The process happens faster and the water holds more salt or sugar when it’s hot.) Sea water then flows through seafloor vents, carrying those dissolved elements up into the ocean’s waters.
Many of those salts and minerals are used by ocean life. Iron, zinc, and copper, for example, are removed from water by organisms. Sodium and chloride, the main ingredients in table salt, are not. Because they are left behind, levels of salt in the ocean have built up over time. This is the reason sea water is salty.
On average, sea water contains about 3.5 percent salt. That salt makes sea water more dense than freshwater. The increased density makes people, animals, and other objects more buoyant in sea water.
The saltiness of sea water (what scientists call salinity) varies across the oceans. It tends to be lower near the equator and the poles. But salinity increases in the areas between. Some seas, like the Mediterranean, are saltier than the rest of the ocean. And some lakes, such as Mono Lake in California and the Caspian Sea in Asia, are saltier still. As water evaporates from these land-locked bodies of water, salts are left behind. Over time, salt levels continue to go up. Many of these salty lakes are in dry areas with limited rainfall and high daytime temperatures.
LEARN MORE
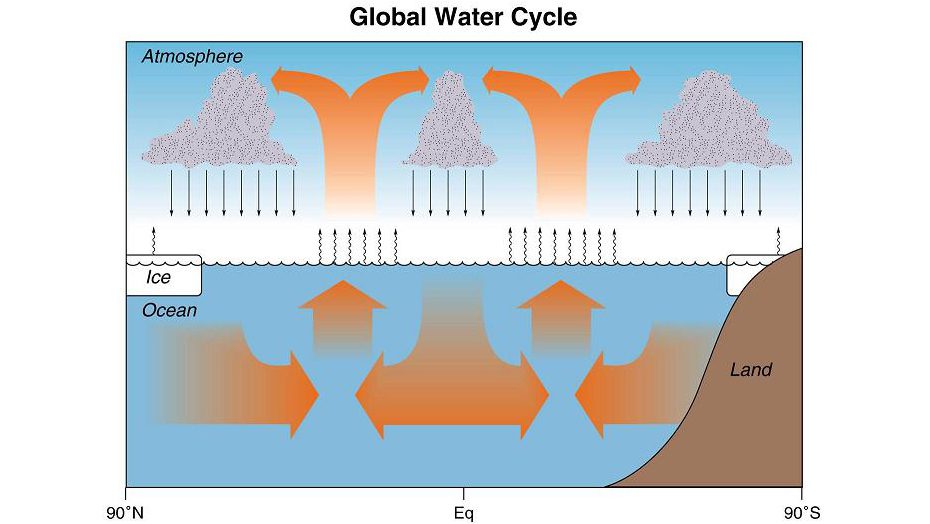
The Global Water Cycle (Dr. Ray Schmidt)
Water Cycle
The water cycle describes the continuous movement of water on, above and below the surface of the Earth.
Hydrothermal Vents
In 1977, scientists made a stunning discovery on the bottom of the Pacific Ocean: vents pouring hot, mineral-rich fluids from beneath the seafloor.
Illinois Department of Physics. Q&A: Salt and buoyancy. https://van.physics.illinois.edu/QA/listing.php?id=16090
NOAA. Why is the ocean salty? https://oceanservice.noaa.gov/facts/whysalty.html
NOAA. Why is the ocean salty, but rivers flowing into it are not? https://oceanservice.noaa.gov/facts/riversnotsalty.html
USGS. Saline Water and Salinity. https://www.usgs.gov/special-topic/water-science-school/science/saline-water-and-salinity
USGS. Why is the ocean salty? https://www.usgs.gov/special-topic/water-science-school/science/why-ocean-salty
Woodward, S.L. Biomes of the World. Department of Geoscience, Radford University. https://php.radford.edu/~swoodwar/biomes/?page_id=1246
DIVE INTO MORE OCEAN FACTS
Are corals plants, animals, or rocks?
The base of a coral reef is coral, but what is coral? If you look at a piece of coral that washed up on shore, it’s solid and tough with rough edges and little pits.
How are seashells made?
One of the most striking features of our beaches is seashells. Their whorls, curves, and shiny iridescent insides are the remains of animals. But where do they come from?
Why do emperor penguins toboggan?
Learn why Emperor penguins slide around on their bellies or “toboggan” when they’re on the move in Antarctica.
What happens in the ocean as a hurricane passes over?
Hurricanes are powerful storms that cause massive damage on land. Here’s a look at what happens below the surface during a storm.