
Mixing Oil and Water
Tracking the sources and impacts of oil pollution in the marine environment
Drop by drop—that is how most oil enters the oceans. Catastrophic spills make the headlines, but it is the chronic dribble, dribble, dribble of seemingly small inputs that supplies most of the oil polluting the world’s oceans.
In recent decades scientists have made substantial progress in understanding how oil enters the oceans, what happens to it, and how it affects marine organisms and ecosystems. This knowledge has led to regulations, practices, and decisions that have helped us reduce sources of pollution, prevent and respond to spills, clean up contaminated environments, wisely dredge harbors, and locate new petroleum handling facilities.
But tracking the sources, fates, and effects of oil in the marine environment remains a challenge for a number of reasons. For starters, oil is a complicated mixture of hundreds, sometimes thousands, of chemicals. Every source of oil, and even the same general types of oil (crude oils or fuel oils, for example) can have distinctive compositions depending on which oil field or well they came out of and how they were refined.
This varying, complex mixture of chemicals gets spilled or seeps into an already complex chemical chowder of seawater, mud, and marine organisms in the ocean. There, the oil is stirred by currents, tides, and waves, altered by other physical processes, and changed further by chemical reactions and interactions with organisms in the sea.
In the midst of this dynamic situation, scientists seek to pinpoint the impacts of oil on myriad individual species, as well as on entire ecosystems. Here the challenge comes full circle, because we now know that the impact of oil can vary greatly, depending on its distinctive chemical composition. But let’s start at the beginning.
How does oil get into the ocean?
Scientists have known since the 1970s that accidents account for only a small percentage of the oil entering our waters. In fact, accidental spills of all types—from ships, shore facilities, pipelines, and offshore platforms—contributed just 9.8 percent of the oil entering the marine environment on an annual, worldwide basis between 1990 and 1999 (but just 3 to 4 percent in U.S. waters).
That doesn’t mean we should dismiss the importance of spills. Accidents such as the 1989 Valdez incident off Alaska or the 2002 Prestige spill off Spain can have devastating effects on marine life and on people’s ability to use the ocean. The impact of a spill depends on the type of oil, the amount spilled, the ocean and weather conditions, and the dynamics of the area or ecosystem where it takes place.
Progress in prevention—through more stringent laws, rules, and guidelines, and increased vigilance by industry and regulators—has reduced accidental spills, at least in developed countries. For instance, studies of tanker spills have prompted regulations for the steady, ongoing replacement of single-hulled tankers in the world fleet with double-hulled tankers.
But spills are just one small way, albeit dramatic, for oil to mix with our waters. So where is the rest of it coming from?
• Seeps: Between one-third and one-half of the oil in the ocean comes from naturally occurring seeps. These are seafloor springs where oil and natural gas leak and rise buoyantly from oil-laden, sub-seafloor sediments that have been lifted close to the earth’s surface by natural processes.
If oil is natural to the oceans and if it is the biggest source of input, what is the fuss about oil as a pollutant? The answer lies in the locations and rates of oil inputs. Oil seeps are generally old, sometimes ancient, so the marine plants and animals in these ecosystems have had hundreds to thousands of years to adjust and acclimate to the exposure to petroleum chemicals. On the other hand, the production, transportation, and consumption of oil by humans often results in the input of oil to environments and ecosystems that have not experienced significant direct inputs and have not become acclimated.
• Extraction: Accidental and normal operation of oil drilling and production platforms puts some oil into the sea, including oil mixed into the briny waters that escape from the oil reservoirs. But this spillage and waste, and the atmospheric releases from platform equipment, is one of the smallest sources of oil in the sea.
• Transportation: In the 1960s and 1970s, scientists studied tar balls collected along major tanker routes and the beaches downstream. They revealed that large amounts of oil were entering the marine environment as a result of ballast water (bilge) discharges and other aspects of “normal” tanker operations. As a result of that research, international conventions and national laws and regulations have led oil shippers to minimize their discharges, particularly in harbors. Today, in spite of increasing numbers of tankers plying the seas, the amount of oil spewed has stabilized or decreased in many places.
• Consumption: The everyday use of oil in cars, trucks, industrial and manufacturing plants, and other machinery of the modern economy is the most egregious and insidious source of oil pollution. The drippings and emissions from millions of machines accumulate on land and eventually run into our waterways.
From the 1950s through the 1970s, one of the most common sources was the indiscriminate disposal of used automobile crankcase oil down sewer systems. Since that time, scientific findings have prompted regulations and public awareness campaigns that promote the recycling and proper disposal of used oil.
But we still have a problem. Peer underneath your parked car and observe the drip of oil. This happens day and night for millions of automobiles, trucks, and buses, creating a chronic, significant source of oil to the sea. Rainstorms wash these drippings into streams or storm sewers that discharge into harbors and rivers. It is terribly hard to measure these types of inputs.
Fossil fuel hydrocarbons from engine exhaust also accumulate in the atmosphere. Sometimes the soot is deposited into our waters; otherwise it is washed out of the atmosphere and into the oceans by rain or snow.
On a smaller scale, new research has shown that outboard engines of small boats and pleasure craft are a significant source of oil pollution—up to 2.2 percent of all inputs in U.S. waters (worldwide data are not available). Engine manufacturers are responding with new models of engines that release much less oil and gasoline, but it may take some time for these changes to propagate through the boating community.
What happens to oil in the ocean?
Oil chemicals entering the ocean have many fates. Volatile chemicals are lost by evaporation to the atmosphere. Other chemicals are broken up by photochemical reactions (catalyzed by sunlight). Bacteria can degrade certain oil components.
The combination of biological, physical, and chemical processes is usually referred to as weathering. These weathering reactions have different rates depending on the chemical structure of the oil, habitat conditions (such as water temperature or oxygen and nutrient supply), and mixing of the water by wind, waves, and currents. In some spills, oil does not last much beyond weeks to months.
But when oil pours into shallow waters with muddy sediments—such as marshes or lagoons—and conditions allow the oil to become mixed into the mud, it will generally persist for a long time. This is a result of the fundamental chemistry of oil compounds. Since they don’t dissolve in water, oil compounds tend to adhere to particles in the water or get incorporated into biological debris, such as fecal matter or dead organisms. These oiled particles and debris settle from the water column and become part of the sediments on the bottom.
Once mired in the sediment, some oil chemicals can persist for years or decades, depending on the environment. In areas swept by high-energy currents, the material may be dispersed. In areas where sediments accumulate (such as ship channels through urban harbors), the contaminated sediments become an environmental concern—both when simply lying on the bottom and when channels are dredged and the mud must be disposed.
For some types of oil inputs and some ecosystems, we now have enough information to model and simulate the fate of oil in the water. This scientific knowledge helps policymakers make better decisions on how to dredge harbors, control pollution sources, clean up contaminated areas, and locate petroleum facilities.
How does oil affect marine life?
From experiments and field measurements, we know that certain types and concentrations of petroleum chemicals can harm marine life. Long-term effects of oil exposure can alter the physiology and ecology of populations of marine organisms, especially those found in sensitive habitats.
Biological and physical processes can reduce the concentration of oil chemicals in an ecosystem, especially if the source of pollution is cut off. As concentrations decline and chemical compositions change, plant and animal communities usually rebound. But the recovery can range from months to decades depending on the chemistry, the conditions, and the organisms and ecosystems affected.
One of the significant advances in the 1970s and 1980s was the development of guides to the sensitivity of various types of coastal ecosystems to oil pollution. Maps of sensitive ecosystems are now used during responses to accidental oil spills, improving the ability of resource managers and engineers to assess where containment booms and other prevention and cleanup measures should be deployed.
There have been few studies, however, on the cumulative effect of chronic inputs of oil to the marine environment, including the many sources associated with oil consumption on land. Assessing these impacts is complicated because oil runoff is often accompanied by other polluting chemicals, making it difficult to tease out which ones have which deleterious effects. Limited experiments have taught us that the interactive effects among chemicals can either increase or decrease each chemical’s long-term effects, depending on the organisms and chemicals.
Much of our knowledge about the effects of oil is still limited. It has focused on biochemical and physiological effects on a few individual organisms and on the degradation of a few particular habitats. But we need a better understanding of the large-scale effects of oil on entire communities and populations, rather than individual organisms. The complexity of how species interact within ecosystems—such as how damage to one species can affect the other species that feed on it—leads to contentious debate whenever regulators start to weigh long-term impacts on marine life.
Fuel for further research
A high priority for the future should be better monitoring and research so we can better quantify how much oil is really entering our waters, how much of it is coming from each source, and what the effects may be.
We also need to expand research on oil pollution in deeper waters. Most concerns and research have traditionally focused on coastal waters. Yet new concerns arise as oil production moves offshore. We can only speculate on the impact of oil exploration and production in deeper waters until we have more detailed knowledge of the biological organisms in these habitats and the biogeochemical processes that govern their lives.
Developed countries and emerging countries consume more oil every year, and that consumption leads to more and more inputs of oil to the oceans. We have learned from our colleagues in developing countries that increased use of fossil fuels has not been accompanied by the sort of stringent regulations developed countries adopted after years of harsh lessons. Further research and education can help those countries minimize the adverse impacts of oil inputs on their oceanic ecosystems.
Editor’s Note: A recent study by the U.S. National Academy of Sciences, entitled Oil in the Sea III: Inputs, Fates and Effects, provided the foundation for this article. To read that report, visit www.nap.edu/books/0309084385/html.
Slideshow
Slideshow
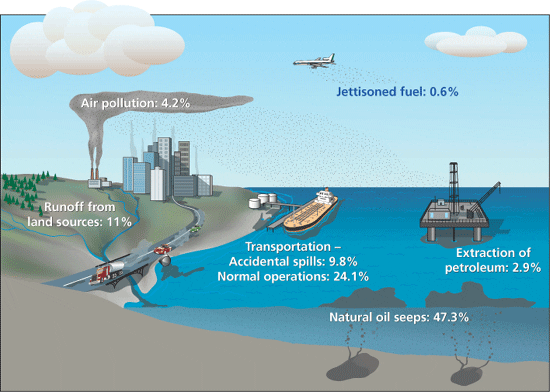
 About 380 million gallons of oil enter the world's oceans and coastal waterways each year from natural and human sources. This illustration shows the approximate worldwide percentages arriving from each source. The relative inputs of oil can vary significantly in different parts of the world. The percentages for transportation include oil lost specifically in oil/petroleum commerce (tankers, pipelines, etc.), as well as the normal operation of all other sea-going vessels.
About 380 million gallons of oil enter the world's oceans and coastal waterways each year from natural and human sources. This illustration shows the approximate worldwide percentages arriving from each source. The relative inputs of oil can vary significantly in different parts of the world. The percentages for transportation include oil lost specifically in oil/petroleum commerce (tankers, pipelines, etc.), as well as the normal operation of all other sea-going vessels.- Satellite radar images show the extent of the 2002 Prestige oil spill along a 30-mile stretch of the northwest coast of Spain. The slick (black areas) is visible because the oil smooths the ocean surface. The surface of clean water appears bright because it scatters and reflects light back towards the satellite. Oil can have wildly different impacts depending on where it is spilled and what the weather is doing in the hours and days afterward. (Courtesy: RADARSAT / Canadian Space Agency.)
- Researchers from the WHOI Marine Chemistry and Geochemistry Department collect samples of oil deposited on the shores of Falmouth, Massachusetts, following an April 2003 spill. Three generations of WHOI scientists have studied the effects of oil in the marine environment through firsthand observations of four nearby spills and smaller, chronic inputs from development and commerce in the industrialized Northeast. (Tom Kleindinst, WHOI Graphic Services.)
- The everyday "normal" activities of modern industrialized society—driving, shipping, heating our buildings, operating industrial machinery—put far more oil and its byproducts into the oceans than all the major spills combined. The significance of the input of oil from petroleum-fueled vehicles might even be understated, since the chronic drip of oil from millions of cars and trucks is extremely hard to measure. (Digital Vision Ltd.)



