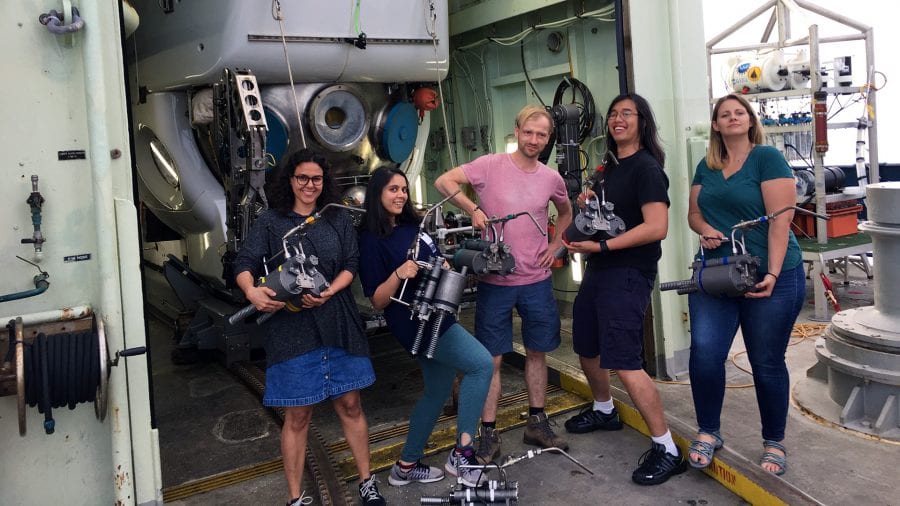Journey to the Bottom of the Sea
Searching for ingredients that may have sparked life on Earth
My eyelids were tightly pressed down as I mustered all the tricks I could think of to get myself to sleep. I rolled around with no sign of getting close to slumber. I had no ticking bedside alarm clock to mark the passage of time. The only audible sound was the thunderous waves crashing against the side of the research vessel Atlantis.
Tonight was far from ordinary. Soon after sunrise, I would dive to the bottom of the Pacific Ocean in the submersible Alvin. I had learned about Alvin for the first time in a biological oceanography class I took in my sophomore year at Chulalongkorn University in Bangkok, Thailand. That seemed like a long time ago. Since then, I had become a graduate student in the MIT-WHOI Joint Program in Oceanography, and I had seen so many photos and videos of hot springs on the seafloor, spewing hot fluids like geysers into the deep ocean. But all of those were nothing compared with what I was about to witness with my own eyes.
My goal for this dive was to collect some samples of hot fluids venting from the seafloor 1.6 miles beneath the surface. I was looking for a particular chemical compound in the fluids: ammonia.
Many of you know ammonia as a common ingredient in cleaning products or fertilizers. But it is also scientifically intriguing, because it is the nitrogen-containing compound that can be converted most easily into key building blocks necessary for life: amino acids—the foundation for proteins and enzymes.
Ammonia is scarce in the ocean, but you can find it in large amounts at vents. Not only are vents oases of life in the deep sea today, they are also thought to be among the possible places where the first life forms on Earth emerged in the distant geologic past. My hope is to study the chemical fingerprints of the ammonia in vent fluids and unravel the chemical processes going on beneath the seafloor that produce it. If we can better understand the geochemical forces that produce ammonia today at vents, it will help open a window into what may have been happening in Earth’s ancient ocean to spark life.
Down to the abyss
Each Alvin dive requires an extraordinary amount of preparation. Many scientists studying various aspects of vents were aboard Atlantis, and Alvin was at the service of all of them. At a meeting the night before my dive, Stefan Sievert, a biologist at Woods Hole Oceanographic Institution and chief scientist of our expedition, went through the long list of tasks to accomplish during the dive. The pilot would execute those tasks, but it is imperative for the scientists inside Alvin to fully understand them, as they often have to make instantaneous judgment calls on how to proceed when questions and unanticipated circumstances arise—as they commonly do.
Now you probably understand why numerous thoughts were racing in my mind as I rehearsed every possible scenario that might happen on the dive and went through everything that needed to get done.
The next morning, full of adrenaline, I got into Alvin. The descent to the seafloor took a little more than an hour. The scene there was eerie; all around us were barren fresh lava rocks. Beyond the vents themselves, not much lives down there except for occasional weird-looking, eel-like fish.
The temperature reading at the seafloor was about 1.6° Celsius (35° Fahrenheit), barely above the freezing point. We started on our long list of scientific tasks to accomplish during the six to eight hours at the bottom.
Crab Spa was the first stop. This is the name of a lukewarm hot spring venting fluids with temperatures of about 25° C (77° F). It hosts numerous tubeworms, mussels, and, as you may have guessed, crabs, which, unlike their colorful shallow-dwelling counterparts, are all white. They spent most of their time around vent orifices that were spewing warm water out of the seafloor. The crabs were trying to grab pieces of white mats made of microbes and shove them into their tiny mouths. It was a pretty amusing scene to witness.
Vent-SID in action
After we took some fluid samples from Crab Spa vents, our next task was to deploy a newly developed instrument called the Vent-SID, short for Vent-Submersible Incubation Device. Sievert and his colleagues are studying the metabolic activities of microbes in vents. To date, scientists have conducted this research by collecting vent fluids, bringing them back on board the ship in incubation devices, and trying their best to simulate the pressure and temperature conditions found at vents.
Investigating these microbial activities through experiments conducted in situ is a better way of understanding what is actually happening in nature. But that’s a daunting challenge. Vent-SID is designed to be placed near vents and suck vent fluids into an incubator chamber. Inside, microbes in the vent fluids carry on their normal biochemical activities, which Vent-SID can measure. The key is that all these chemical reactions are carried out in situ, under the conditions in which they normally happen.
Finding a perfect spot to place the 3.5-by-2.5-by-8-foot-tall Vent-SID was not easy. Yet Pat Hickey, a veteran Alvin pilot with more than 680 dives, did not take long to use Alvin’s two manipulator arms and put Vent-SID right above a Crab Spa vent orifice. Then he maneuvered a temperature probe into the venting fluids to find the best place for the Vent-SID to take fluids into its incubator chamber.
Vent-SID would automatically start filling the chambers with vent fluids (along with the microbes in them) and begin its job as an in situ robotic lab, performing experiments scientists typically do either onboard the ship or back in a lab on land—including injecting chemical tracers, collecting microbes after incubation, and preserving samples for genetic materials. The experiments will help elucidate the kinds of microbes down there and the types and rates of biochemical reactions they perform—in an effort to understand the critical roles microbes play in sustaining vent ecosystems.
Chimneys on the seafloor
We had several more vent sites to visit on our dive. Our next stop was a spectacular site called Bio9. When we approached the area, the clear water was suddenly filled with what looked like dense black smoke. Then it was as if that velvety black curtain was pulled open, and we could see several enormous structures that looked like rocky chimneys spewing the black smoke. These tall structures form when the hot vent fluids meet cold seawater and almost instantly solidify. The “smoke” spewing out is actually vent fluid filled with minerals dissolved from subseafloor rocks. Some of the chimneys were extinct, and others were actively emitting very hot fluids.
Black-smoker chimneys form when the vents fluids are hot. Fluids emanating from Bio9 can be as hot as 360° C (680° F). The fluids and inner chimney walls are too hot for microbes, as life can tolerate temperatures up to only 122° C (252° F). But the fluids cool down quickly when they spew into seawater, creating a more hospitable environment for life to thrive.
Fluids from high-temperature vents such as Bio9 are also particularly important for my research, because they are too hot for living things, such as microbes. So any ammonia in them cannot have been produced by chemical reactions involving microbes. That is the type of ammonia that billions of years ago could have been converted into amino acids that led to the first life on Earth.
We proceeded to collect fluid samples from Bio9 with what we call Major samplers. We had a lot of samples from high-temperature vent sites. So we called it a successful day, dropped Alvin’s weights, and floated back to the surface.
A hard day’s night
My long day in the submersible was no excuse for not having a long night in the lab.
My colleagues and I worked as fast as we could in clockwork fashion to get the vent fluids out of the samplers and into appropriate bottles and then process and store them away either in the refrigerator or freezer.
These samples were particularly foul-smelling. Every time we cracked open the Major samplers to take out the vent fluids, the smell of sulfide permeated the room. It is the same rotten-egg smell you experience from muddy salt marshes. This sulfide, as well as the ammonia that I measure, provide the chemical energy that vent microbes use to live and grow.
But this repugnant smell strangely reminded me of the childhood summers I spent at my grandparents’ home. In their backyard stood a dense mangrove swamp reeking of the same sulfide smell. My life seemed to have come full circle.
My teammates and I carefully extracted fluids from the Major samplers, one by one. We measured the fluids’ pH and salinity—the first line of evidence to confirm the integrity of the samples we had collected.
Most low-temperature vent samples have a salinity similar to seawater. Their pH, a measure of how acidic they are, ranges between 5 and 6, like that of black coffee. On the other hand, high-temperature vent fluids from the black smokers in this area tend to be fresher and have a pH of 3 to 4, like that of vinegar used in cooking.
It’s the high-temperature fluids that I am after.
Isotopes as a fingerprinting tool
Work does not end at sea. Back in the lab at WHOI, we analyze our precious samples with an isotope ratio mass spectrometer (IRMS), hoping to reveal the unique chemical fingerprints hidden within compounds found in the vent fluids. These chemical fingerprints are isotopes—forms of chemical elements with different masses.
I study nitrogen isotopes—more precisely, the ratios between the heavier and scarcer nitrogen-15 to the lighter and more abundant nitrogen-14. Scientists have used these nitrogen isotope ratios for decades to study food webs, trace fertilizer pollution in watersheds, or study the many chemical reactions involving nitrogen that happen in the ocean.
With my Ph.D. advisor, WHOI biogeochemist Scott Wankel, I am investigating the nitrogen-isotope fingerprints to reveal what chemical processes are happening below the seafloor to give rise to ammonia in hydrothermal vent fluids.
I also worked with WHOI geochemist Jeff Seewald to set up closely controlled lab experiments to learn more about ammonia-producing processes. I conduct these reactions within a bag made of gold. Gold is malleable and can withstand high pressure and heat, and it does not react with the other chemicals. So it makes a fine container to put in our reactor at high temperatures and pressures similar to those at seafloor vents.
There are many chemical pathways that can produce ammonia. Which is the major one? What conditions of pressure, heat, rocks, geological features, or other factors favor this pathway versus another? What scenario is more likely to make ammonia? What’s going on at present that would let us infer what might have been happening in ancient oceans to spark the emergence of life?
Like any problem in science, understanding the origin of ammonia in vent fluids is not straightforward. We are just beginning to uncover the geochemical processes beneath the seafloor that produce ammonia. Analyses of fluids from vents in different oceans may show other scenarios beyond what we see at Bio9 and other sites in the Pacific.
The more I explore the subject, the more I find questions begging for answers. And just as during the sleepless hours before my Alvin dive, these scientific questions from time to time keep me awake at night.
This research was funded by the National Science Foundation. Net Charoenpong is supported by the NSF, the J. Seward Johnson Fund, and a Thailand Ministry of Science and Technology fellowship.
From the Series
Slideshow

Slideshow
- Chawalit Charoenpong, a graduate student in the MIT-WHOI Joint Program, uses a blowtorch to heat up a bag made of gold in preparation for lab experiments to learn more about how ammonia is produced at deep-sea hydrothermal vents. Gold is malleable, can withstand high pressure and heat, and does not react with the other chemicals. So it makes a fine container to put in a reactor at high temperatures and pressures similar to those at vents. (Tom Kleindinst, Woods Hole Oceanographic Institution)
- The manipulator arm of the human-occupied submersible Alvin takes a sample of fluids from Crab Spa, a low-temperature hydrothermal vent in the Pacific Ocean. Could vents have provided key chemicals that laid the foundation for life to arise on an ancient Earth? (Courtesy of Stefan Sievert, WHOI/NSF/HOV Alvin)
- Net Charoenpong (second from right) and his colleagues happily retrieve samplers that pilots aboard Alvin used to collect samples of fluids from seafloor vents. (Andrew Babbin, MIT)
- Vent-SID (Submersible Incubation Device) sits near an active hydrothermal vent on the seafloor. It draws vent fluids into an incubator chamber and measures the metabolic activity of microbes in the fluids—under pressure and temperature conditions in which they normally occur. (Courtesy of Stefan Sievert, WHOI/NSF/HOV Alvin)
Related Articles
- The story of a “champion” submersible
- Robots to the Rescue
- Who is Alvin and what are sea trials?
- 7 Places and Things Alvin Can Explore Now
- The story of “Little Alvin” and the lost H-bomb
- Meet the Alvin 6500 Team: Lisa Smith
- Overhaul to take Alvin to greater extremes
- Meet the Alvin 6500 Team: Rose Wall
- Racing an undersea volcano
- The Discovery of Hydrothermal Vents
- Pop Goes the Seafloor Rock
- Bringing a Lab to the Seafloor
- ‘Covering’ Alvin‘s History
- Alvin‘s Animals
- Alvin‘s Fun Facts
Topics
Featured Researchers
See Also
- Follow the Trails of Nitrogen Through Hydrothermal Vents
- The Human-occupied Vehicle Alvin
- Scott Wankel
- Jeff Seewald
- An Ocean Instrument Is Born An article on the Submersible Incubation Device from Oceanus.
- Bringing a Lab to the Seafloor An article on the Submersible Incubation Device from Oceanus.