Hidden Battles on the Reefs
How will corals fare in a changing ocean?
How do you drown a coral reef?
The very idea seems unfathomable for animals that spend their entire lives under water. But the deep ocean is actually riddled with “drowned” coral reefs—the remains of ancient reefs that slipped into the dark ocean depths and starved without sunlight.
To stay afloat in the ocean, people work hard to keep their heads just above water. Corals do the opposite, striving to stay just under the surface of the ocean. Coral reefs need to get it just right—submerged in the sea, but shallow enough for the corals’ symbiotic photosynthetic algae to soak up sunlight. Too deep and the ecosystem wastes away without solar energy to make food. Too shallow and corals dry out at low tide.
This delicate balance is achieved by a constant tug-of-war, which most people overlook. Day in and day out, as corals build their skeletons up toward the sea surface, other organisms are eroding and dissolving the skeletons to build their own homes, cutting down the corals’ hard work.
Today, the chemistry of our ocean is rapidly changing. How sensitive are corals and bioeroders to these changes? How will changing ocean conditions favor or thwart each side in this battle? Will they tip the delicate balance and expand the graveyards of drowned coral reefs?
An undersea tug-of-war
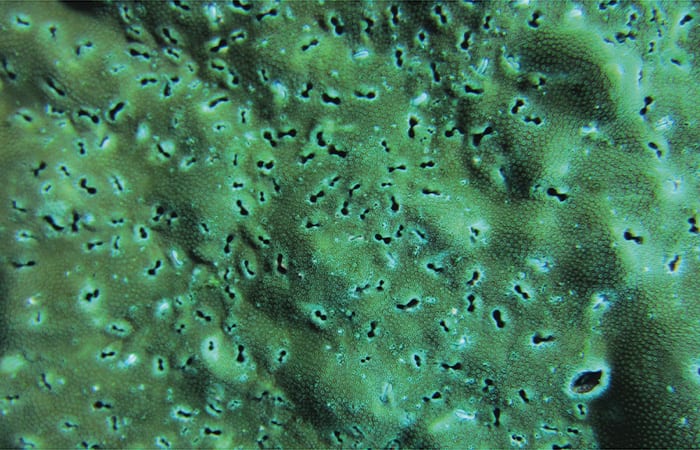
Corals make their skeletons out of calcium and carbonate ions dissolved in seawater, constructing massive structures as large as your car, maybe even your house. But at the same time, a collection of other organisms called bioeroders—mollusks, worms, and sponges—bore into the skeleton of living corals to find shelter, each leaving distinctly shaped tunnels or borings in the reef framework.
Corals usually win this war, keeping the reef near the sea surface and within reach of sunlight. But if the balance is tipped and the reef becomes eroded down far below the sea surface, there is no longer enough sunlight for corals to survive, and they will “drown.”
This delicate balance between calcium carbonate production and removal is threatened by ocean acidification, the decline in seawater’s pH driven by rising carbon dioxide (CO2) levels in the atmosphere. Not all of the CO2 emissions from our cars and factories stay in the atmosphere. The ocean has already absorbed about a third of it. As CO2 enters the ocean, a well-known series of chemical reactions takes place and carbonate ions, the currency of coral reefs, begin to disappear. Reducing the amount of carbonate ions available in seawater for corals makes it harder for them to build their skeletons up to the surface. It’s like putting ankle weights on people makes it harder for them to stay afloat.
Flattened reefs
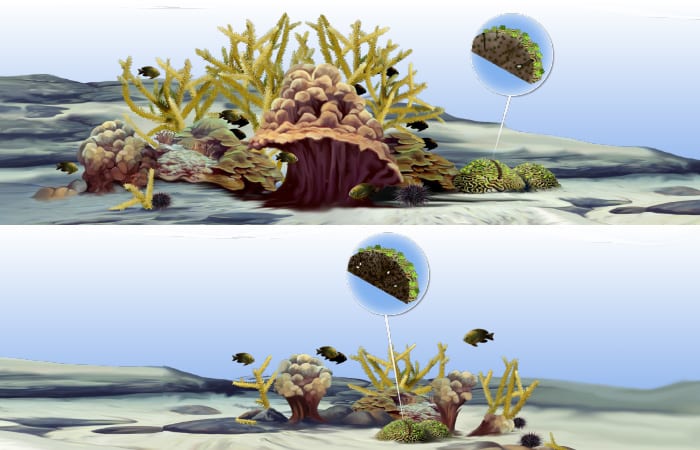
When corals and bioeroders are in harmony, the former grow just fast enough to stay near the sea surface, while the latter are busily sculpting the coral skeletons into an intricate, three-dimensional habitat full of nooks and hiding places for the more introverted creatures that call reefs home.
We generally picture coral reefs inhabited by creatures of every color imaginable, with reef fish and sharks swimming about. But a closer look reveals just how many more creatures are hiding within the reef.
When I scuba dive on reefs, I spend most of my time swimming a few feet above the bottom looking down. During a recent expedition to the U.S. Virgin Islands, I realized I was completely missing the hidden underworld. One of my jobs was to count adult and juvenile urchins—a baseball-size creature with foot-long needle-like spikes sticking out in all directions. Urchins usually try to hide, but adults never seem to find a hiding place quite big enough and their spines stick out in plain view. But I had never seen a juvenile urchin.
To find one, I poked my head in all the crevasses of the reef. In every nook, there was some creature hiding out: juvenile urchins, shy reef fish, and mean-looking groupers with sharp teeth. Swimming above the reef, I might see one or two urchins along a 30-foot line, but I counted more than 30 once I looked closely. (Sticking one’s head in holes on a reef is not always a great idea, though. One hiding spot was the home of a particularly inhospitable five-foot long Moray eel, which came shooting out of its hole with jaws full of sharp teeth snapping.)
In a lower-pH ocean with less carbonate ions available, corals may not be able to build new skeleton fast enough to keep pace with bioeroders cutting down the reef. The result would be flatter coral reefs with less of the three-dimensional structure that makes these ecosystems so vibrant. Because many reef fish depend upon this complicated structure to hide, flatter reefs may threaten the amazing biodiversity found on coral reefs.
A trans-Pacific quest
Changing ocean conditions are bound to affect the bioeroders, too. Will lower-pH seawater hinder them, or will it help them and tip the balance even further against corals?
As a member of Anne Cohen’s lab at Woods Hole Oceanographic Institution (WHOI), I’ve been investigating the impacts of ocean acidification on coral reefs. Our team has searched the ocean for reefs that are able to tolerate living in seawater that naturally has a pH lower than the seawater typically found in the tropics. This search took us from the Pacific coast of Panama, across a vast stretch of the equatorial Pacific Ocean, and into the South China Sea—just shy of half the circumference of the globe.
Some of these reefs are among the most remote places in the world. In satellite images, they are odd sand-colored pixels. To seafarers, they are dull piles of sand and a few palm trees, and to cartographers, they are annoying specks. Traveling to and working on these remote reefs is not easy. A bunch of wooden planks tied to eight plastic pipes passes for a research vessel. Yet, just below the sea surface, these reefs are underwater jungles teeming with life.
CAT scans and computers
We use underwater drills to collect cores of skeleton from wild coral colonies. Coral colonies can be several meters tall and wide, yet only the very surface—the width of your little finger or less—is living. Below the living coral lies the calcium carbonate skeletons built by the colony over tens or hundreds of years.
Inside the skeletons, the bioeroders are undoing the careful architecture of the coral. The cores we collect give us a peek into this otherwise hidden battlefield.
Precisely measuring bioerosion in our cores is where things get really tricky. The borings in the skeletons are all different shapes and sizes. Some borings make a hole all the way through a core; others are completely hidden inside. Clams and worms keep their homes neat, carving out smooth holes in the skeleton. Sponges are messy, building their homes in all sorts of haphazard shapes with apparently little rhyme or reason. Rather than trying to measure the sizes of the borings myself, I trained a computer to do it for me.
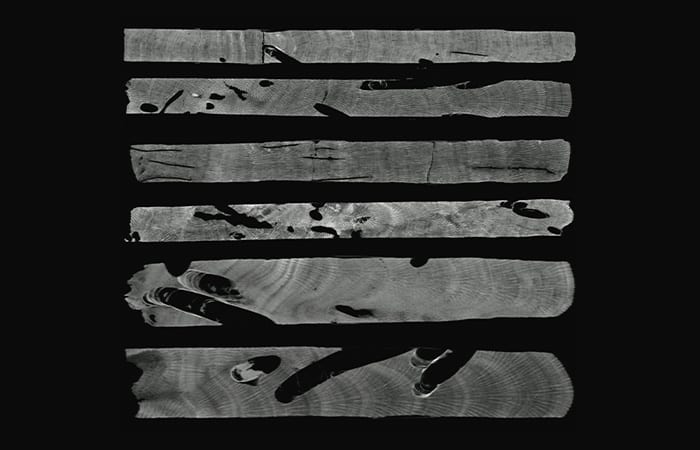
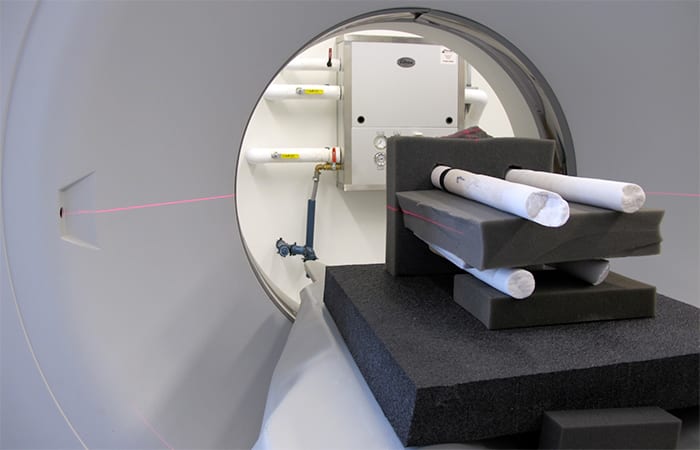
All of our cores go through a CAT scanner at the Computerized Scanning and Imaging Facility here at WHOI, similar to one you would find at a hospital. Computerized Axial Tomography technology gives us a 3-D picture of the cores, even inside the skeleton where we cannot see.
The precision of this technology is incredible, resolving at approximately the width of a human hair. But this precision comes with a price: A two-foot long core has close to a billion data points. That’s where a computer comes in handy—all these data were crunched in a few minutes to measure how much skeleton the bioeroders had removed.
On our search across the Pacific, we identified a number of reefs that are naturally happen to be bathed in seawater with pH levels today that are as high as those expected for much of the tropical ocean by the end of this century. Examining the 3-D CAT scans of coral skeletons collected from these reefs, we found that relatively acidic (lower-pH) reefs were more heavily bioeroded than their higher-pH counterparts.
A tipping balance
But there is another factor at work. The bioeroders responsible for most of the destruction of coral skeleton are filter feeders. They sift particles of food out of seawater. The ultimate source of the food is phytoplankton, the grasses of the ocean at the bottom of the food chain, but corals also feed on the zooplankton, the tiny animals that are one chain-link above the phytoplankton.
In places where there are plenty of nutrients in the ocean, plankton grow quickly, supplying food for large populations of bioeroders. But these nutrient-rich regions also tend to be relatively acidic, making it difficult to tell whether bioeroders are affected by nutrients, pH, or both.
On our search across the Pacific, we identified naturally occurring coral reef ecosystems that live in settings that span a range of different pH and nutrient conditions in the ocean. That allowed us to investigate the isolated and combined influences of acidification and nutrients on the bioerosion rates of coral skeleton.
Our most striking finding was that in waters with a combination of high nutrient levels and lower-pH, bioerosion is ten times higher than in lower-pH waters without high nutrient levels. Our study was published online Nov. 14 in the journal Geology.
So how do you drown a reef? The answer is “add acid,” and to do it much faster, add nutrients. Both are happening now on many of our coral reefs.
The ocean will certainly absorb more CO2 over the next century, and ocean acidification is a global phenomenon that reefs cannot escape. But the encouraging news in our findings is that people can take action to protect their local reefs. Most excess nutrients added to the ocean come from human activities that cause runoff from sewers, septic tanks, roads, and fertilizers. If people can limit these sources of nutrients to the coastal ocean, the bioeroders will not have such an upper hand, and the balance will tip much more slowly toward erosion and dissolution of coral reefs.
This research is funded by the National Science Foundation (NSF), the Nature Conservancy, and the National Oceanic and Atmospheric Administration’s Coral Reef Conservation Program. Tom DeCarlo is supported by an NSF Graduate Research Fellowship.
From the Series
Slideshow
Slideshow
- Graduate student Tom DeCarlo, right, has sailed the world studying coral reefs, from the Caribbean Sea to the South China Sea. (Hanny Rivera, Woods Hole Oceanographic Institution)
- To study corals in the South China Sea, WHOI scientists set up a makeshift floating lab, strapping wooden planks across plastic tubes. (Kathryn Pietro, Woods Hole Oceanographic Institution)
- A makeshift boat carries equipment and the research team out to the coastal coral reefs that they will dive on to sample and study. From left, Tom DeCarlo, Kathryn Pietro, and Pat Lohmann of Woods Hole Oceanographic Institution, and Kristen Davis from the University of California, Irvine. (Lisa Hou, Academia Sinica)
- On all coral reefs, an unseen tug-of-war is going on. While corals grow toward the sunlit surface, organisms called bioeroders—mollusks, worms, and sponges—bore into coral skeletons to find shelter. The bioeroders also sculpt nooks and crannies in the skeletons that create hiding places for other creatures. If all is in balance, the coral reef becomes a vibrant, diverse habitat (top). But if factors hinder coral growth or give bioeroders the upper hand, coral reefs will become flattened and die (bottom. (Illustration by E. Paul Oberlander, Woods Hole Oceanographic Institution)
- This coral off the coast of Panama shows holes bored into its skeleton by bioeroders. (Hannah Barkley, Woods Hole Oceanographic Institution)
- The research team has been studying this relatively acidic coral reef in the Palauan archipelago. Seawater pH on this reef today represents acidification levels predicted for tropical western Pacific by the end of the 21st century. (Tom DeCarlo, Woods Hole Oceanographic Institution)
- To study a coral, Tom DeCarlo uses an underwater drill to extract a thin core of its skeleton. The drill hole is sealed with cement so that the coral can continue to grow. In about a year, the coral will have grown completely over the hole, leaving no trace of our sampling. (Anne Cohen, Woods Hole Oceanographic Institution)
- The CT scan images have a resolution of about the width of a human hair. The images show the borings made by bioeroders in the coral skeleton. (Tom DeCarlo, Woods Hole Oceanographic Institution)
- The scientists used the Computerized Scanning and Imaging Facility at WHOI to get CT scans of their coral skeleton core samples. (Tom DeCarlo, Woods Hole Oceanographic Institution)
Related Articles
- Cold, quiet, and carbon-rich: Investigating winter wetlands
- The 10,000-foot view
- A cascade of life
- An introduction to marsh bothering
- A toxic double whammy for sea anemones
- Squid Games
- Waves of inspiration
- Burrows on the beach
- Where the Rivers Meet the Sea
- Marshes, Mosquitoes, and Sea Level Rise
- The Unseen World on Coral Reefs
- The Recipe for a Harmful Algal Bloom
- Life at the Edge
- The Bacteria on Your Beaches
- Reassessing Guidelines for Oil Spill Cleanups
Featured Researchers
See Also
- Anne Cohen's Lab
- Coral macrobioerosion is accelerated by ocean acidification and nutrients Study published in Geology magazine
- A Quest for Resilient Corals Oceanus magazine
- Palau's Merand Blogs & Expeditions
- Expedition to Dongsha Atoll Blogs & Expeditions
- Tom DeCarlo's Homepage
- MIT-WHOI Joint Program in Oceanography
- Computerized Scanning and Imaging (CSI) Facility at WHOI