Having Their Phosphorus and Eating It Too
Plankton's unusual ability may give it an ecological edge
Try naming the nutrients you need to be strong and healthy. Chances are phosphorus will be pretty far down the list, if it’s there at all.
And yet, you and every other organism on Earth—including those in the ocean—need phosphorus. It’s an essential chemical brick in DNA’s double-helical scaffold, and it’s the “P” in ATP, or adenosine triphosphate, the basic molecule that most cells use for energy.
“Phosphorus is a critical macronutrient required for all life in the sea, and especially for phytoplankton,” the tiny marine plants at the base of the food chain, said Sonya Dyhrman, a biologist at Woods Hole Oceanographic Institution (WHOI). Half of all the photosynthesis on the planet occurs in the ocean (mostly by phytoplankton), and since the process requires phosphorous, the nutrient’s importance is obvious.
But until recently, little attention was paid to oceanic phosphorus, partly because it was so hard to get reliable measurements of the phosphorus in seawater.
When new techniques for detecting minute amounts of the nutrient became available about 15 years ago, they revealed that the ocean contains far less phosphorus than most ecosystems on land. Dyhrman and other scientists began to wonder: Does the availability of phosphorus in the ocean help determine where marine life can flourish? Could the relatively barren regions in the north Atlantic and north Pacific be due to a shortage of phosphorus?
An unlikely source
Can any marine organisms use phosphonates? Dyhrman asked. In 2006, a team of scientists including Dyhrman and former WHOI biologist Eric Webb found that several species of a phytoplankton called Trichodesmium have a special set of genes encoding the machinery that enables them to break the phosphonate bond and get the phosphorus out. The researchers found that Trichodesmium use more accessible forms of phosphorus if they’re available; but if those forms are scarce, whether in a lab culture or in a nutrient-poor region of the North Atlantic, Trichodesmium turn on the genes that allow them to acquire phosphorus from phosphonate.
Dyhrman said that ability likely gives Trichodesmium an advantage over their competitors in areas of the ocean where phosphorus is in very short supply. But it raises another question: Where are the phosphonates coming from? Levels of phosphonates are the same in nutrient-rich and nutrient-poor regions of the ocean; if Trichodesmium in nutrient-poor areas are breaking down phosphonates, what is renewing the supply?
Dyhrman and marine geochemist Claudia Benitez-Nelson of the University of South Carolina thought the most likely source was one or more species of plankton. To find out if any common marine microbes make phosphonates, the scientists used nuclear magnetic resonance spectroscopy to measure various chemical forms of phosphorus, including phosphonates, in dozens of single-celled species. In test after test, they detected little or no phosphonates in the cells—“until we started looking at Trichodesmium,” Dyhrman said.
Cornering the market
In a study published in the October 2009 issue of Nature Geoscience, she and her colleagues reported that in one species, Trichodesmium erythraeum, from 8 to 17 percent of the phosphorus was in the form of phosphonates. “That is more phosphonate than anyone has found in any kind of microbe, and it’s certainly the first significant detection of phosphonate in a marine microbe,” she said.
The results were astounding: The same species can not only use phosphonates, it can also produce them.
“It is a little confounding, but it could be a clever strategy,” Dyhrman said. “There are some other microbes that can use phosphonate, but not many. So the Trichodesmium erythraeum is basically creating an environment where it’s self-selecting for itself. You can imagine this scenario would give it a competitive advantage.”
That advantage is likely to come into play even more in years to come, as climate change affects the marine environment, Dyhrman said. “In the future ocean, we expect Trichodesmium erythraeum to grow really well. It likes warm temperatures, and it does well under high CO2 [carbon dioxide] levels. That suggests it’s potentially going to be producing more phosphonate in the future.”
Which would be good news for the few other organisms that can use phosphonates, including other species of Trichodesmium. It could also be good news for the oceans’ productivity and their ability to store carbon, because all the Trichodesmium species have yet another special chemical ability: They can convert atmospheric nitrogen into compounds that other organisms can use, much as legume plants do on land. In effect, Trichodesmium provide nitrogen “fertilizer” that boosts the ability of other phytoplankton to photosynthesize and take up more CO2.
Dyhrman cautioned that the news may not be all good, depending on the specific phosphonate compounds that T. erythraeum makes. If those compounds are methylated—that is, if they contain a methyl (CH3) group—then when they are broken down to release their phosphorus, they will also release methane—an even more potent greenhouse gas than CO2.
This research was supported by the National Science Foundation, the Center for Microbial Oceanography Research and Education, the Princeton Center for BioInorganic Chemistry, and the Ocean Life Institute of the Woods Hole Oceanographic Institution.
Slideshow

Slideshow
 WHOI biologist and microbial oceanographer Sonya Dyhrman. (Photo by Tom Kleindinst, Woods Hole Oceanographic Institution)
WHOI biologist and microbial oceanographer Sonya Dyhrman. (Photo by Tom Kleindinst, Woods Hole Oceanographic Institution)- WHOI biologist Sonya Dyhrman (left) and MIT/WHOI Joint Program student Elizabeth Orchard lower a sampling net from the research vessel Atlantic Explorer into the waters of the Sargasso Sea, a nutrient-poor region of the North Atlantic. Dyhrman, Orchard, and their colleagues found that Trichodesmium in this region turn on the genes that allow them to acquire phosphorus from phosphonates, a skill few other marine organisms have. (Photo by former WHOI postdoctoral fellow Alena Sevcu)
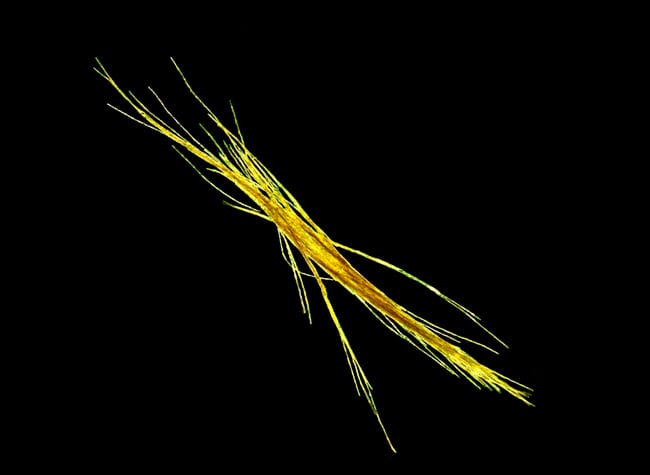
- Species of the single-celled phytoplankton Trichodesmium form colonies with distinctive shapes. Individual colonies, shown here, are visible to the naked eye; where currents and winds gather many colonies together, the aggregation can be seen from orbiting satellites. (Photo by MIT/WHOI Joint Program student Abby Heithoff)
- A colony of Trichodesmium erythraeum. All species of Trichodesmium can obtain phosphorus from phosphonates, but so far only T. erythraeum has been found to produce phosphonates. (Photo by MIT/WHOI Joint Program student Abby Heithoff)
Related Articles
- The little big picture
- Are warming Alaskan Arctic waters a new toxic algal hotspot?
- The Living Breathing Ocean
- Forecasting Where Ocean Life Thrives
- PlankZooka & SUPR-REMUS
- Illuminating an Unexplored Undersea Universe
- Specks in the Spectrometer
- Setting a Watchman for Harmful Algal Blooms
- Short-circuiting the Biological Pump
- A Telescope to Peer into the Vast Ocean
- Jet Fuel from Algae?
- New Use for Well-known Algae
- Trouble in the Tropics
- Proteomics Reveals Ocean’s Inner Workings
- What Causes the Atlantic to Bloom?
Featured Researchers
See Also
- A Most Ingenious Paradoxical Phytoplankton From Oceanus magazine
- Little Things Matter a Lot From Oceanus magazine
- The Many Faces of Trichodesmium Slideshow from Oceanus
- A Leg Up for Tiny Trichodesmium Blog post from the field
- Ocean Microscope Reveals Surprising Abundance of Life About Cabell Davis and the video plankton recorder from Oceanus magazine
- Mystery Solved: Marine Microbe is Source of Rare Nutrient Press release on Dyhrman's recent discovery