The Living Breathing Ocean
Can ‘ocean elevators’ carry excess carbon to the deep?
Rainforests have been dubbed the Earth’s lung, but like us, our planet has two lungs. The second one is the ocean. Rainforests and oceans both draw in carbon dioxide from the atmosphere, reducing the buildup of heat-trapping gas that is warming our climate. Both use photosynthetic plants to capture carbon dioxide from the atmosphere and convert it into organic carbon that the plants use to grow. But while one of these lungs relies on large trees that grow slowly over decades, the other involves microscopic organisms capable of surviving no more than a few weeks every year. Two very different pathways, producing essentially the same result: regulating the amount of carbon dioxide in Earth’s atmosphere.
The tiny organisms in the ocean are called phytoplankton. Do not let their short life expectancy and small size fool you. Though they are no wider than a human hair, phytoplankton serve the same purpose as the giant kapok trees found in the Amazon. And there are billions and billions of them. Think of it as crowdsourcing: If every phytoplankter captures even a little bit of carbon dioxide, the total effect can be very large.
Nearly 25 percent of the carbon dioxide in our atmosphere is “pumped” into the ocean, where it converted into organic carbon by phytoplankton at the ocean surface all over the world. But a lot remains unknown about the fate of this carbon once it has penetrated into the ocean. Some of it goes up the food chain, some of it dissolves into the ocean, some of it is released back to the atmosphere, and some of it sinks into deeper parts of the ocean. This last pathway is especially interesting. If the carbon sinks deep enough, it potentially could remain in the ocean for thousands of years. This one-way ticket is a promising way to efficiently “bury” carbon deep down and keep it from returning to the atmosphere.
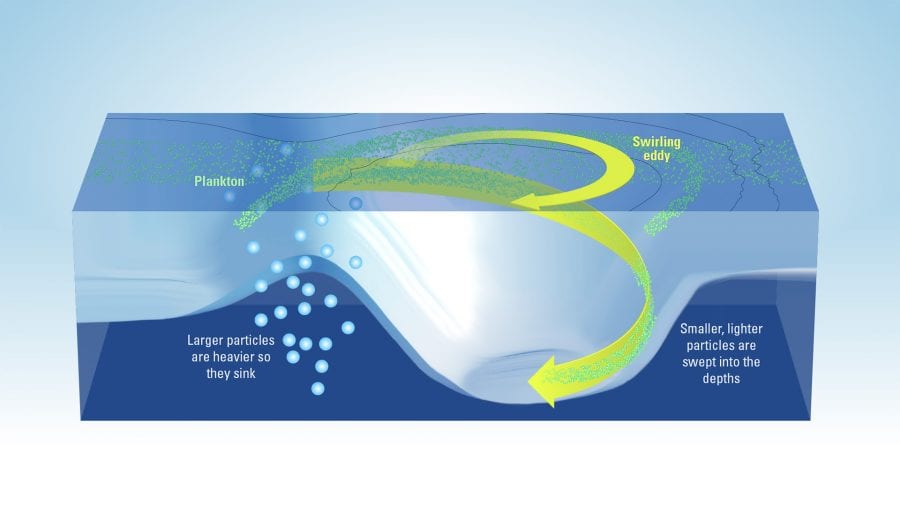
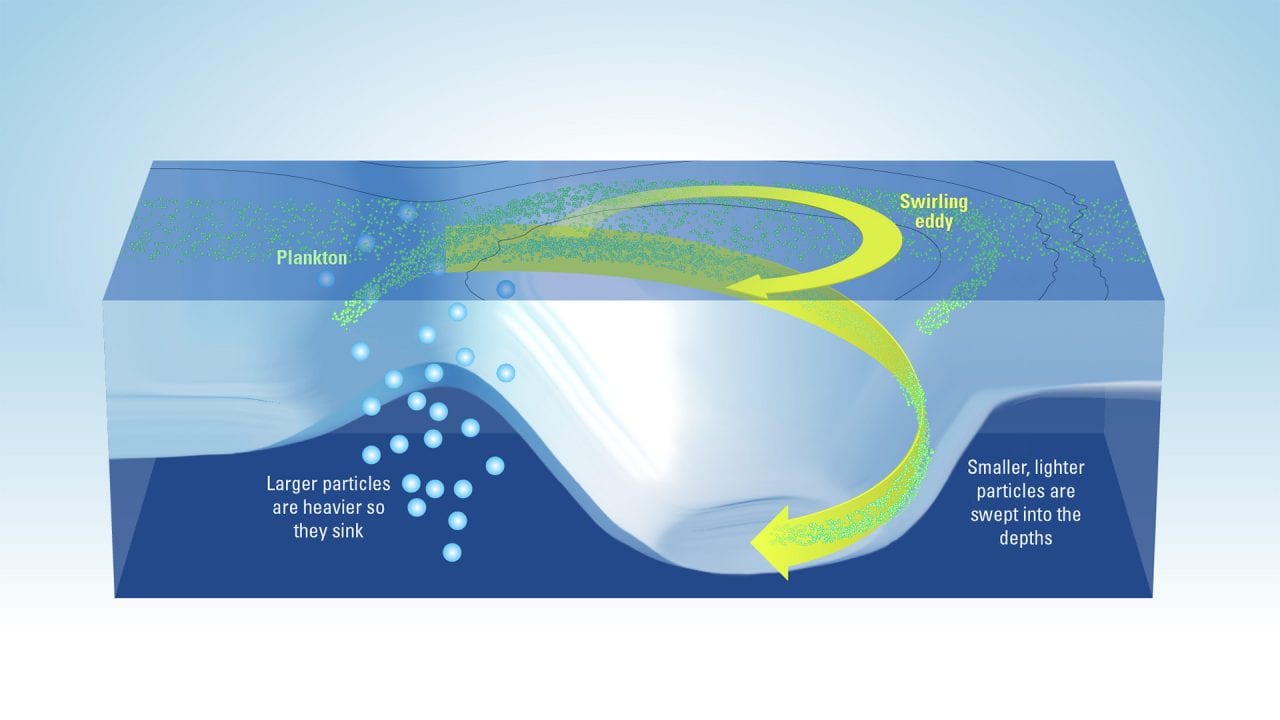
This process is often called “carbon sequestration,” and scientists have traditionally thought that it is achieved when particles—dead organisms or fecal pellets, for example—are large and heavy enough to sink out of the ocean surface to greater depths. Smaller and lighter particles, despite being far more numerous, generally float near the surface, being stirred around by ocean currents until they decompose. Scientists have therefore assumed that these smaller particles don’t contribute to carbon sequestration into the ocean.
But what if they did? What if they contributed quite a lot?
Underwater fireworks
You can think about what happens in the ocean as fireworks. After they explode, a few large shining sparks fall all the way down, while most of the smaller sparks fade away before they get a chance to reach the ground. In the ocean, when conditions are right, the phytoplankton population explodes, creating a large number of particles of all sizes. Heavier particles sink fast enough to transport the carbon they contain to greater depths before they biodegrade. On the other hand, lighter particles disappear before they get a chance to sink deep enough.
Well, to be honest, this is only mostly true. This paradigm has been widely accepted for the main reason that ocean currents travel mainly in the horizontal direction. Generally, it’s harder for water to move up and down, and so far less water moves vertically, and when it does, it moves very slowly—about a hundred times slower than a garden snail’s top speed!
However, scientists have recently discovered that, through complex processes and in very localized regions, vertical currents do exist, and they can be much larger than we originally had thought and measured. We were missing them up to now. Why? Because those stronger vertical currents are too localized to be seen from satellites and too ephemeral to be measured consistently by oceanographers at sea.
What goes down must come up
More and more, the scientific community agrees that this water moving vertically in the ocean can act as an elevator capable of carrying light particles from the surface to greater depths, including particles that would otherwise be too light to sink on their own. Though these smaller particles each contain less carbon than larger ones, these particles are far more numerous and therefore could significantly contribute to transferring carbon from the surface to deeper depths.
This has shaken our understanding of how the ocean can regulate the amount of carbon dioxide in the atmosphere, as well as our capability to predict how the ocean will respond to the ever-increasing buildup of carbon dioxide in the atmosphere. These vertical elevators suggest that we have been underestimating how much carbon dioxide these previously disregarded smaller particles can contribute to removing carbon from the atmosphere and sequestrating it in deeper layers of the ocean.
To improve our understanding of this phenomenon, we must address many unanswered questions. How and where do these elevators form in the ocean? What happens to particles as they are moving down? How fast do those vertical elevators have to move to ensure that even small particles make it deep into the ocean before they biodegrade? Can we quantify how much more carbon sinks when those vertical elevators are present, compared with when they are absent?
This is where things get even more complicated. In the ocean, what goes down must come up—meaning that if some water is taking an elevator down, some equal amount of water must be taking another elevator up to compensate. And these going-up elevators would contain a hard-to-distinguish mixture of both larger and smaller particles.
We now have two competing mechanisms for carbon sequestration: When these vertical oceanic elevators are not present, larger particles (sparser but high in carbon) all sink deep down, and smaller particles (much more numerous but low in carbon) are all biodegraded and never make it to the deep ocean. In places where the elevators are active, many more smaller particles are sent to the deep ocean, but some larger particles are sent back up, so fewer make it down.
So what is the net effect? Are those elevators really enhancing the total amount of carbon exported into the deep ocean? And, most importantly, how can we figure it out?
Observational challenges
Because vertical currents are both localized and short-lived, it is difficult to observe them directly in the ocean. It’s also a great challenge to measure and assess the sizes and distribution of all the particles in the water.
NASA has recently put together an impressive expedition to make field observations of particles sinking and try to correlate them with satellite observations of the surface ocean. The program is called EXPORTS (Export Processes in the Ocean from Remote Sensing), and it involves scientists from many institutions, including Woods Hole Oceanographic Institution.
Meanwhile, a team of scientists at WHOI—including me, Amala Mahadevan, and David Nicholson—has been using a numerical ocean model. This advanced computer program aims to reproduce properties of the real ocean (currents, temperature, salinity, etc.) as well as processes, including those that form vertical currents. In the comfort of our offices, we can add in imaginary particles with different sizes and sinking speeds and see what happens to them as time passes. Our three main questions are:
When, where, and why do larger vertical currents occur in the ocean?
How do these vertical currents interact with particles of all sizes in the ocean?
Do vertical currents enhance or reduce the amount of carbon sequestered into the deeper parts of the ocean?
Our preliminary results have revealed that horizontal and vertical ocean currents are closely linked. In regions where horizontal currents are very strong, the water flow tends to become unstable, creating swirls of spinning waters at the currents’ peripheries. Those swirls, spinning downward like underwater hurricanes, generate vertical currents in the ocean. Think of the swirling water that appears above the drain hole in your bathtub.
Our computer model was able to recreate those swirls. Then we added particles to the model to see what happens. Our computer simulations revealed that the vertical currents are indeed very efficient at exporting small particles to the deep ocean. Not only did they transport a large amount of small particles downward, they also moved them much more rapidly than we had anticipated. Does this increased export of small particles to the deep have a greater net impact after accounting for the resurfacing of large particles? That is something our team is still actively working to resolve.
These insights, if confirmed, would significantly change our understanding of the ocean’s role in regulating carbon dioxide in the atmosphere. We may have been underestimating it. Over the past few decades, we have gained a good understanding of the oceanic lung in the Earth’s respiratory system. Our attention now has to zoom down to the level of “oceanic alveoli”—those smaller features that might very well play a fundamental and heretofore overlooked role in our climate system.
This research was funded by the National Aeronautics and Space Administration.
From the Series
Image
Slideshow
- Plankton in the ocean can help reduce the amount of heat-trapping carbon dioxide building up in our atmopshere. Like plants, they use carbon dioxide to grow and are eaten by larger organisms. Large, heavy particles sink to the depths, carrying down carbon so it can’t return to the atmosphere. Scientists are investigating whether large amounts of smaller, lighter particles can be swept into the depths by swirling eddies. (Illustration by Eric S. Taylor, WHOI Creative Studio/based on illustration by Melissa Omand, University of Rhode Island)