Let There Be Laser Light
Laser spectroscopy could illuminate key environmental gases
Earth’s warming climate is poised to cause changes on our planet. In the Arctic, permafrost has begun to thaw, releasing methane, a heat-trapping greenhouse gas, into the atmosphere. On the seafloor, warmer ocean temperatures threaten to release methane gas by thawing solid deposits of methane hydrates found in cold depths.
Such changes are potentially momentous, but how can we detect evidence that they are happening? How can we measure tiny trickles of gases wafting over enormous and remote areas?
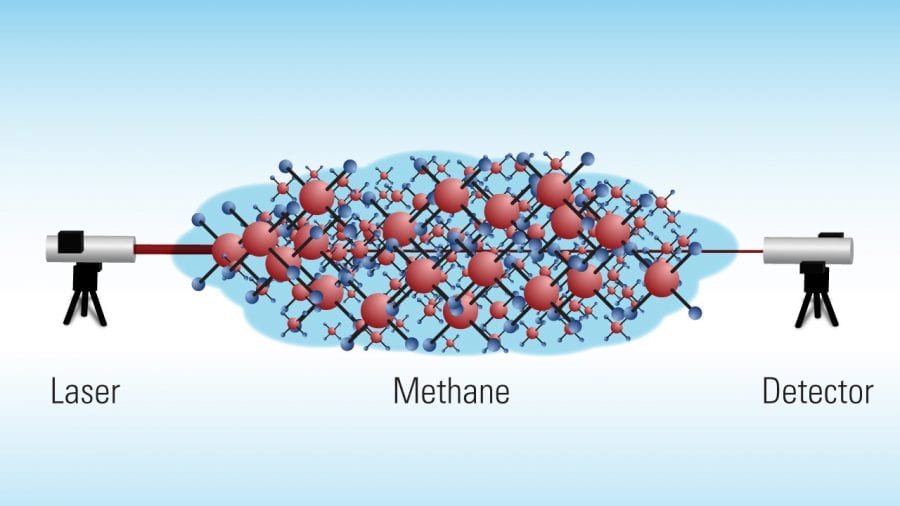
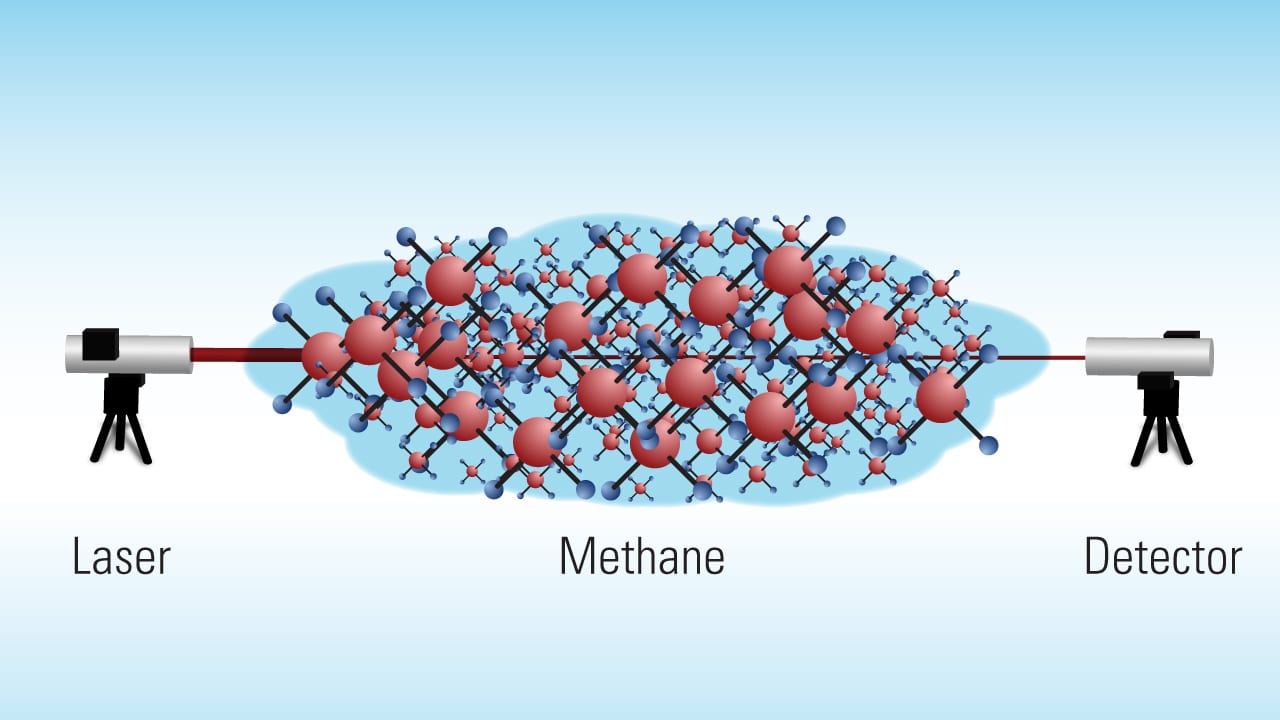
One answer may lie in using optical methods, says Anna Michel, a scientist in the Applied Ocean Physics and Engineering Department at Woods Hole Oceanographic Institution (WHOI). Michel is engineering new devices that use a technique called laser spectroscopy. Essentially, it shoots a laser beam through air or water to a receiver, measuring the amount of light that emerges at the far end. In between, different chemical compounds will absorb distinct wavelengths of light.
“If more of the targeted gas is present in the air or water, less light gets through on the other side,” Michel said.
Laser spectroscopy can be used to make highly sensitive measurements of a variety of trace gases in the air, such as methane and carbon dioxide. Among its other distinct advantages, laser spectroscopy is non-invasive and doesn’t disturb the environment, and it allows scientists to “sense” chemicals right at the scene.
“Instead of having to take a sample and bring it back to the lab, we want to be able to do it in the field,” Michel said. “We also want to be able to make measurements over long-distance scales. If you take one measurement in one location, you may not be getting a full understanding of what’s happening.”
Michel is also developing laser spectroscopy to work in a completely different medium—water, rather than air—so it can be used under the sea. She’s pursuing instruments that function autonomously, and she’s exploring ways to make devices smaller and cheaper, so that more can get out into the field. With the help of collaborators at WHOI—geochemist Scott Wankel and engineer Jason Kapit—she’s well on the way to overcoming the engineering challenges to make these advances possible.
A long path to success
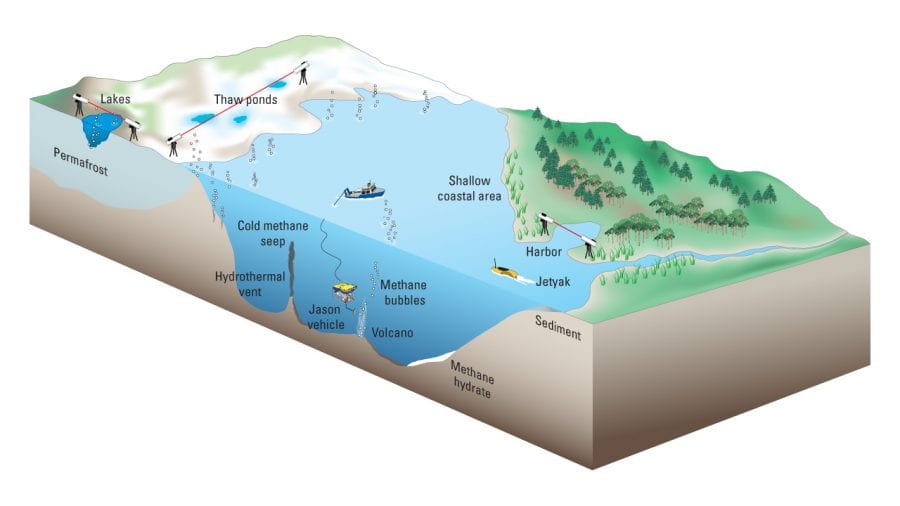
Laser spectroscopy would provide a great tool for the Arctic, where vast fields of frozen soil, or permafrost, hold huge amounts of organic matter that can thaw and release unknown amounts of methane. To date, scientists have used “point sensors” to take measurements in only one place at a time, making it hard to capture the whole picture of what is happening.
Methane emissions are often concentrated in “hot spots” that are easy to miss or overvalue with point sensors, Michel explained. To capture methane emissions across a wide area, she and her colleagues are developing a long-path sensor that can measure gases across kilometer-scale expanses. It includes two telescopes, one containing the laser and the other the detector. A data-acquisition system coupled to the detector measures and records the amount of light that travels between the two instruments.
Michel and Kapit tested the system to measure gas emissions across Cape Cod ponds. “Batteries in the system will be hooked up to solar panels and potentially wind turbines, which will give them the power to run for long periods of time,” Kapit said.
Bringing the lab to the seafloor
Next, the scientists want to take laser spectroscopy under the ocean, where gases are dissolved in water.
“We’re taking something that should work in the air and now you want it to work in water and in this high-pressure environment,” Michel said.
She is collaborating with Scott Wankel, an isotope biogeochemist at WHOI, to modify a laser Wankel originally developed during his postdoctoral research at Harvard with Peter Girguis to measure gases dissolved in fluids and bubbles under water.
“What we analyze is a gas, so we need our samples to be in its gas form,” Wankel said. The device diffuses seawater through a Teflon membrane that extracts gas from the water. In gas form, the compounds absorb laser light.
Among their scientific questions, Michel and Wankel want to understand how much methane emerges from undersea volcanoes, hydrothermal vents, and seafloor deposits of methane hydrate—a frozen compound that incoporates methane in a solid form but can thaw and return methane into a gas. They also want to know how much methane reaches the surface.
“The more we look, the more we see these methane bubbles coming out of the seafloor,” Wankel said. “Are there more methane bubbles today, or do we just have better ‘eyes’?” he said, referring to improved ways to observe the phenomenon.
The scientists are also interested in other chemical compounds in the ocean, including the greenhouse gas carbon dioxide. Detecting carbon dioxide is even more complicated, because some is converted to other compounds, such as calcium carbonate, in seawater. To reverse the chemical changes, the scientists added a pump to the membrane that acidifies seawater to convert all sources of carbon back into carbon dioxide.
“By measuring under water with laser spectroscopy, we get more points faster, in real time, with much higher data density,” Wankel said. “We could go for two weeks and come back with twenty samples, while one dive with a laser spectrometer could get forty to fifty points.”
Taking the first dive
In 2014, Michel and Wankel tested their underwater laser spectroscopy instrument on a remotely operated undersea vehicle that measured carbon dioxide bubbles from Kick’Em Jenny, an underwater volcano 590 feet below the surface off the coast of Grenada. It is the most active volcano in the Caribbean.
The researchers took the same instrument to another curiously named seafloor feature: the “Jacuzzi of Despair” in the Gulf of Mexico. It is a pool of water so salty and dense that it does not mix with overlying seawater. Few organisms that venture into the Jacuzzi return, and measurements from the spectrometer reveal why: The brine pool is about five times saltier than seawater and contains high concentrations of methane.
“Getting to see something work at the bottom of the ocean is pretty exciting,” Michel said. “A lot of the research we’re doing is really cutting-edge. The technology is proven in atmospheric environments, but there are new ways to make these things better.”
Self-governing sensors
One way is by making the technology more self-sufficient. The current underwater system requires scientists to be on a ship above it, watching every move. But with autonomous technology, the researchers might not need to be there at all.
“I could sit here on my computer, and in theory I could have an instrument out in a deep-sea environment and tell it what to do,” Michel explained.
The researchers have already been toying with this idea. When the spectrometer was tested at Kick’Em Jenny, Michel was actually at her laptop in Rhode Island, watching undersea action on a remote monitor, while Wankel supervised everything on the ship.
“Telepresence allows you to connect with large numbers of people,” Wankel said, “but it’s a developing technology. We were the guinea pigs in that study.”
Michel and colleagues are also working on a laser spectrometer system for a Jetyak, an autonomous motorized kayak that can go on pre-programmed data collection missions.
Getting a handle on things
The researchers also want to make the sensors smaller and cheaper—two important steps that will increase the number of spectrometers that can be used regularly.
The first step is shrinking the size of the sensors by doing more efficient gas extractions. While the lasers and detectors are pretty tiny, the length of the path the gas needs to travel to be detected is large. That means the apparatus has to be large, too.
“The laser spectrometer we have right now is so big I can’t even lift it,” Michel said.
“There’s only so many sensors out there, so we have very limited spatial and temporal measurements,” Michel said. “If we can get to a point where we can make lots of these sensors that can stay out for long periods of time, then we’ll have a much better understanding of some of these processes going on in the environment.”
Is the methane released from Arctic permafrost increasing? Does carbon dioxide output from undersea volcanoes contribute to ocean acidification? How much excess carbon from fossil fuel burning can the ocean really absorb from the atmosphere? Developing more functional atmospheric and deep-sea sensors will be key to answering these critical questions.
This research was supported by the NOAA Office of Ocean Exploration and the Paul G. Allen Family Foundation and by collaboration with the Ocean Exploration Trust.
Slideshow
Slideshow
- The long-range laser spectroscopy system developed by WHOI scientist Anna Michel and colleagues is designed to measure atmospheric levels of methane across long distances. The laser shoots out a beam. Some of the light is absorbed by gas molecules in the air. The more gas present in the air, the less light is detected at the far end. (Illustration by Eric Taylor, WHOI Graphic Services )
- Scientists at WHOI are developing laser spectroscopy systems that can measure critical gases that provide evidence of changes occurring in the environment. They are building systems that can take measurements over long distances—across lakes or thawing Arctic permafrost fields, for example. They are testing systems that can work under water to measure methane and other gases from hydrothermal vents, cold seeps, thawing methane hydrates, and volcanoes. And they are adapting these types of sensors to operate on autonomous vehicles such as the JetYak and remotely operated vehicles such as Jason. (Illustration by Jack Cook, WHOI Graphic Services)
- WHOI scientists are developing an underwater laser spectroscopy system that can be used to measure gases dissolved in fluids and bubbles. The device was designed to measure how much light is absorbed by compounds in air, so the compounds must be in gaseous form for the device to work under water. To extract the compounds as gases, the device passes seawater past a membrane. (Photo courtesy of the Ocean Exploration Trust)
- In 2014, a funnel was used at Kick' em Jenny, a highly active underwater volcano in the Caribbean to collect bubbles for analysis with a laser spectrometer. The researchers were measuring the isotopic composition of carbon dioxide bubbles emerging from the volcano. (Photo courtesy of the Ocean Exploration Trust)
WHOI scientists deployed a deep sea laser spectrometer in a brine pool in the Gulf of Mexico, known as the "Jacuzzi of Despair," in 2015. The Jacuzzi is about five times saltier than overlying seawater and contains high concentrations of methane.
(Photo courtesy of the Ocean Exploration Trust)A hose and reel system was used to take fluid samples from the "Jacuzzi of Despair" brine pool in the Gulf of Mexico. The sampling line was lowered from a remotely operated vehicle (ROV) to a depth of 19 meters in the brine pool. This allowed the researchers to make in situ measurements of fluids in the Jacuzzi.
(Photo courtesy of the Ocean Exploration Trust)