A Change Has Come in the Arctic
A rise in radium in the ocean signals changes along the coast
I was about fifteen minutes into my nap when I heard the announcement: “Polar bear. Port beam. One hundred yards. Huge.”
I paused to consider: Would this be worth losing some of the only precious moments of sleep I could steal before I had to return to the lab? But it didn’t take long to decide. I extracted myself from my bunk aboard the Coast Guard icebreaker Healy and—slowly, to avoid hitting my head again—and threw on as many wool layers as I could find. Was my camera in my room, or in the lab? The lab, I decided, and made my way there, begrudgingly getting in my daily arm workout as I opened and closed each of the ship’s heavy metal watertight doors in my path.
By the time I got outside, the deck was crowded with spectators, scientists and crew alike. The polar bear did not disappoint. She cautiously investigated the ship for the better part of an hour, sniffing the air and rolling around on the ice, before ambling off toward the white horizon. Now I was definitely too excited to go back to sleep.
Arctic wildlife sightings highlighted my emails home, but they weren’t the reason I had undertaken this 65-day journey on the Healy. The Arctic is a unique ocean, and it’s also uniquely susceptible to climate change. Temperatures are rising twice as fast in the Arctic as they are in the rest of the world. Sea ice is already noticeably diminishing, and that is causing far-flung environmental effects across the region.
Climate change could alter the very chemistry of the Arctic Ocean. That’s what I was aboard the Healy to study, and what I discovered was unexpected: The chemistry of Arctic waters is already rapidly changing. These chemical changes indicate that large-scale changes are occurring in how the ocean interacts with the land along the coasts. They signal changes that potentially could affect the Arctic food chain, from microbial life right up to polar bears.
An ocean unlike any other
On typical world maps, the Arctic Ocean stretches across the top, where it forms a thin blue border around the Northern Hemisphere. At a glance, it looks similar in size to the Atlantic and Pacific Oceans. But in fact, the Arctic is quite different from its southern siblings.
It’s a tiny ocean, with only three percent of the volume of the Pacific. It’s also surrounded by land, and that land extends far out beneath the ocean surface to form vast shallow continental shelves.
Shelves cover more than half the area of the Arctic Ocean, and that’s what makes this ocean my dream study location (well, notwithstanding the freezing temperatures and 24-hour darkness in winter). Why? Because the continents and continental shelves surrounding the Arctic Ocean have significant impacts on the ocean’s chemistry.
Rich layers of sediments accumulate atop continental shelves. The sediments are chock-full of elements and chemical compounds that are released and dissolved into overlying seawater. The process is almost similar to steeping teabags, but instead of adding flavor to the water, shelf sediments release chemicals.
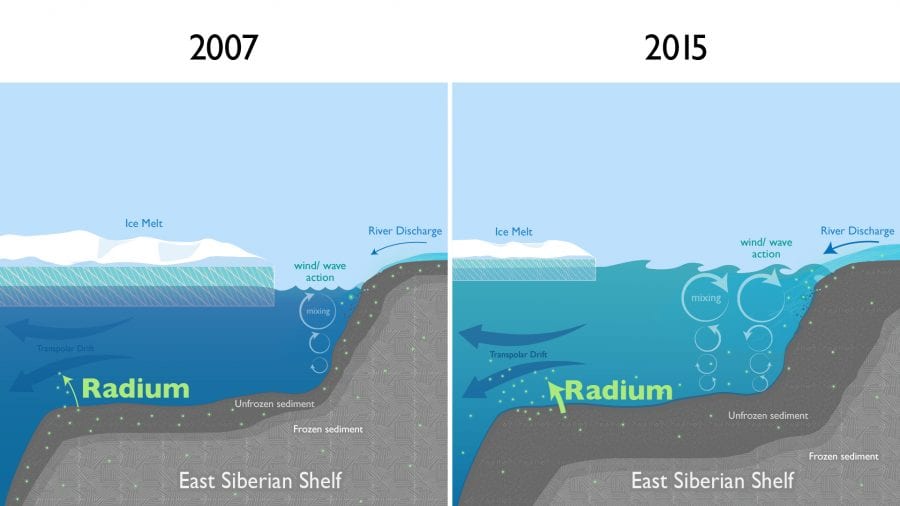
In this process the coast and shelves and ocean all interact with one another in complicated ways. But things may be becoming more complicated because of climate change. Melting sea ice creates more open water near the coast for winds to create waves over shallow Arctic shelves. These more turbulent seas can intensify the erosion of coastlines. Rougher waters reach down and stir up the sediments on shelves, so that more chemicals are released and carried up and away into the ocean by currents.
To predict how these shifts will affect the chemistry of the Arctic Ocean, we need to measure these complex interactions and find ways to monitor changes. That isn’t easy to do, especially over large shelf areas. But, luckily, I had just the tool for the job.
Thanks, Marie Curie
Radium is a naturally occurring radioactive element discovered in 1898 by Marie Curie. All soil, rocks, and sediments contain radium. It also dissolves in salty water, so ocean waters pick up radium when they wash over continental shelves or brush against coastlines. That makes radium an excellent tracer to track land-based materials in the open ocean. We can track where it came from, and determine how long ago it left the shelf.
If we find high levels of radium in seawater in the open ocean, it tells us that the water touched the shelf recently. It also allows us to trace other important chemical compounds that likely were also added to the ocean from shelf sediments at the same time as the radium. These include essential nutrients that are carried up to the surface and sustain life there.
Radium was my element of choice, but many scientists were aboard the Healy to study a wide range of chemicals. The cruise was part of the international GEOTRACES program, aimed at measuring elements and chemical tracers in the world’s oceans to understand their current distributions and to provide a baseline to assess future chemical changes.
Over our two-month voyage in the summer of 2015, I collected samples along the Healy ’s path through the Bering Strait, across the Chukchi Shelf, and through the Western Arctic all the way to the North Pole (where, of course, I paused to take a picture with the iconic red and white striped pole).
To measure the radium in Arctic waters, I used a pump to pull seawater up a hose draped over the side of the Healy. I filtered the water through acrylic fibers that were coated with manganese, which makes the fibers turn black, so they look like gorilla hair. The manganese acts like a sponge. It soaks up the radium from seawater as the water passes through the fibers.
The icebreaker stopped in 69 locations along the way, and at each sampling station, I concentrated the radium from hundreds of liters of seawater onto a bundle of fiber about the size of your cell phone. The fibers were then shipped back to the lab in Woods Hole to be analyzed.
I was surprised by what I found.
Surprising signals from the shelf
Because continental shelves are the source of radium, I expected to find the highest radium levels over the Chukchi Shelf. But when I analyzed my samples, I realized that there was actually more radium in the central Arctic, near the North Pole.
I had expected the radium levels I observed in 2015 to be similar to those previously measured by other scientists in 2007. I was surprised to find that the amount of radium in surface waters near the North Pole had drastically increased over this time period. The levels we measured in 2015 were twice as high as those measured in 2007.
What explained this dramatic increase? And what brought all that radium way out into the middle of the ocean?
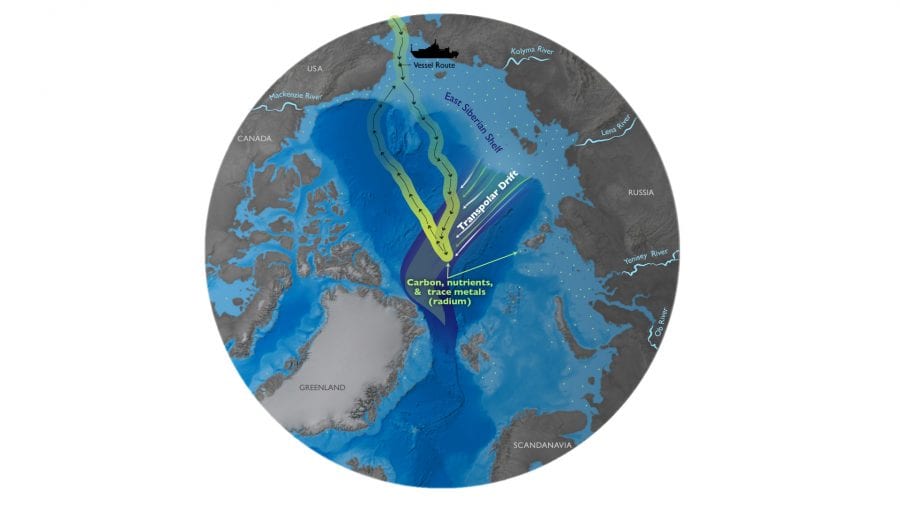
To answer the second question, I scrutinized a few maps of water circulation in the Arctic and quickly fixated on the Transpolar Drift, a strong current that carries water from the continental shelves north of Russia across the central Arctic and eventually through the Fram Strait near Greenland. I realized that the high levels of radium in the middle of the Arctic Ocean were indeed coming from a shelf—just not the one we had crossed on our way out to the central Arctic!
These Russian shelves are in a region of the Arctic experiencing some of the strongest warming. Rapidly rising air and sea temperatures are increasing the number of ice-free days over these shelves. The loss of sea ice allows for stronger wind and wave action, which could increase erosion on the coast and turbulence over the shelf—and thereby add more radium and other materials from sediments to the ocean.
The dramatic increase in radium in the central Arctic Ocean indicates that these large-scale coastal changes—less sea ice, more turbulence, and more dissolved chemicals transported from the shelves into the open ocean—are already happening.
The big picture of tiny organisms
Radium does not play a role in sustaining life in the ocean, but it comes from the same continental shelf sources as the nutrients and metals that nourish tiny marine organisms living in sunlit surface waters. Just as people require iron in hemoglobin to transport oxygen in our blood, marine organisms need small amounts of metals to keep their bodies functioning.
The increased inputs of radium to the central Arctic imply that there must also be increased inputs of other elements, including ones that are important to organisms.
The tiny plants and animals living in Arctic surface waters are already trying to adjust to the loss of sea ice, which is increasing the amount of sunlight they receive. Changes in the amounts of essential nutrients coming from continental shelves would also force adjustments. Some species might flourish in waters with higher nutrient and metal concentrations, but others that are better adapted to living in nutrient-poor conditions could become scarce.
It is important to understand and monitor these shifts because changes that affect species at the bottom of the food chain eventually will have significant impacts on a wide range of Arctic organisms, from shrimp to narwhals to humans.
This ultimately is what motivated me and the other scientists onboard the Healy. The Arctic is a difficult place to do field work, but not many locations offer glimpses of the Northern Lights, polar bear sightings, and interesting chemistry! I am eager to return and learn more about this unique ecosystem. Just let me find my wool layers first.
The National Science Foundation funded this research. Kipp is also supported by a National Defense Science & Engineering Graduate Fellowship.
From the Series
Slideshow

Slideshow
- Lauren Kipp, a graduate student in the MIT-WHOI Joint Program in Oceanography, led efforts to measure radium-228 in the Arctic Ocean. The naturally occurring isotope is used to track the flow of material from land and sediments into the open ocean. Here, she removes a cartridge from a sampling instrument that pumps seawater through the cartridges to collect chemical isotopes. (Cory Mendenhall, U.S. Coast Guard)
- Aboard the icebreaker Healy, MIT-WHOI graduate student Lauren Kipp measured seawater chemistry across the Arctic Ocean and found that levels of radium-228 have almost doubled over the last decade in the middle of the ocean. The radium was transported from land and shallow continental shelves by currents such as the Transpolar Drift. The surprising finding is evidence that rapid climate change is causing large-scale changes along the Arctic coast, such as diminishing sea ice. These coastal changes, in turn, could also deliver more nutrients, carbon, and other chemicals into the Arctic Ocean and have significant impacts on the Arctic food web. (Natalie Renier, WHOI Creative Studio)
- Diminishing sea ice near the Arctic coast leaves more open water for winds to create waves. The increased wave action reaches down and stirs up sediments on shallow continental shelves, releasing radium and other chemicals that are carried up to the surface and swept away into the open ocean by currents such as the Transpolar Drift. A new study found surprising evidence that climate change is rapidly causing coastal changes in the Arctic that could have significant impacts on Arctic food webs and animal populations. (Natalie Renier, WHOI Creative Studio)
- Scientists deploy a pump into the Arctic Ocean that can filter more than 1,000 liters of seawater over four hours. Instruments collect chemicals and particles from the seawater as it is pumped through the device. Scientists measured seawater chemistry from the western Arctic Ocean to the North Pole. (Cory Mendenhall, U.S. Coast Guard)
- Occasional sightings of polar bears are among the bonuses of conducting research in the Arctic Ocean. (Cory Mendenhall, U.S. Coast Guard)
- MIT-WHOI graduate student Lauren Kipp's research took her across the Arctic Ocean, and since she was in the neighborhood, she paid a visit to the ceremonial pole at the North Pole. (Erin Black, Woods Hole Oceanographic Institution)
Related Articles
- Our eyes on the seafloor
- 4 Potential Solutions for Corals in Crisis
- The hypoxic reef
- WHOI scientists discuss the chemistry behind Sri Lanka’s flaming plastic spill
- An ocean of opportunity
- The ocean has heartburn. Is relief on the way?
- Science RoCS Initiative responds to need for increased ocean monitoring
- A new ocean soundscape
- Putting the ‘nuclear coffin’ in perspective
- Tracking Radium in the Arctic
- Meet the ChemYak!
- Chasing Ocean ‘Snowflakes’
- To Tag a Squid
- How Do Corals Build Their Skeletons?
- Searching for ‘Super Reefs’
Featured Researchers
See Also
- Increased fluxes of shelf-derived materials to the central Arctic Ocean From Science Advances
- USCGC Healy
- GEOTRACES
- Polar bear visits the USCGC Healy YouTube video