Small Drop in pH Means Big Change in Acidity
The key danger factor is an increase in dissolved hydrogen ions
Estimated reading time: 2 minutes
FEATURED IN: "The Socioeconomic Costs of Ocean Acidification"
One of the most common negative responses Sarah Cooley gets when she speaks to community groups about ocean acidification is, “What do you mean, ocean acidification? The ocean is not acidic! Seawater is never going to get below pH 7—so you must not know what you’re talking about.”
That’s partly true, said Cooley, a postdoctoral researcher at Woods Hole Oceanographic Institution. The pH of seawater is near 8, which makes it mildly alkaline, or basic; but any decrease in the pH of a liquid is considered “acidification.”
“It’s a lot easier to say ‘ocean acidification’ than ‘ocean de-alkalinization,” said Cooley.
pH is an index of how many protons, or hydrogen ions (H+) are dissolved and free in a solution. The pH scale goes from 0 to 14. A fluid with a pH of 7 is neutral. Below 7, it is acidic; above 7, it is alkaline.
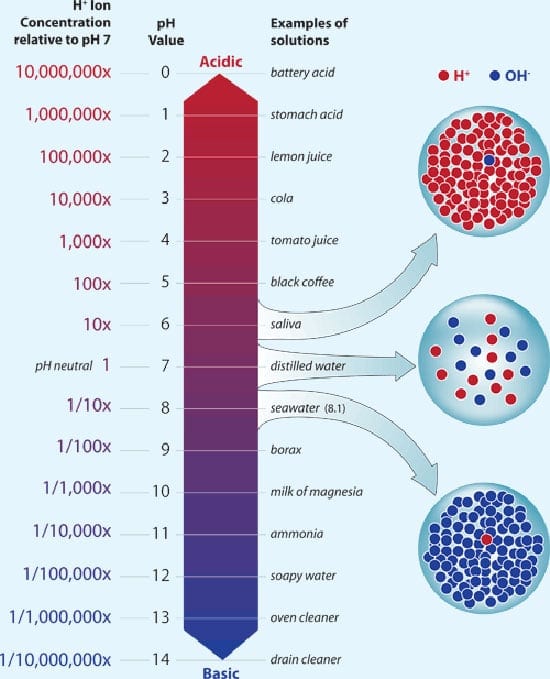
A small drop in pH means big change in acidity: The pH of seawater is near 8, which makes it mildly alkaline, or basic; but any decrease in the pH of a liquid is considered “acidification.” The key danger factor is an increase in dissolved hydrogen ions.(Woods Hole Oceanographic Institution)
A small drop in pH means big change in acidity: The pH of seawater is near 8, which makes it mildly alkaline, or basic; but any decrease in the pH of a liquid is considered “acidification.” The key danger factor is an increase in dissolved hydrogen ions.(Woods Hole Oceanographic Institution)
The more below or above 7 a solution is, the more acidic or alkaline it is. The scale is not linear—a drop from pH 8.2 to 8.1 indicates a 30 percent increase in acidity, or concentration of hydrogen ions; a drop from 8.1 to 7.9 indicates a 150 percent increase in acidity. Bottom line: Small-sounding changes in ocean pH are actually quite large and definitely in the direction of becoming less alkaline, which is the same as becoming more acidic.
If you think about it, we use descriptive words like this all the time. A person who stands 5’5” tall and weighs 300 pounds isn’t thin. If he loses 100 pounds, he still won’t be thin, but he will be thinner than he was before he went on the diet. (And we are more likely to comment that he’s looking trimmer than to say he’s not as fat as he used to be.)
It’s the same with ocean acidification. Seawater is not acidic, nor is it ever likely to be; but because of the buildup of CO2 in our atmosphere, more CO2 is absorbed by the oceans. That makes them more acidic than they used to be—and a lot more acidic than is healthy for corals, clams, oysters, and many other organisms that make their shells or skeletons out of calcium carbonate.
The increased acidity doesn’t corrode the shells and skeleton, per se; rather the excess H+ ions bond with carbonate ions to make bicarbonate, leaving fewer carbonate ions available for organisms to use in shell-building and requiring a greater outlay of energy by the calcifying organisms.
The origin of the term “pH” is unclear. The “H” stands for hydrogen ions; the “p” has been suggested to mean either “power” (so pH would mean “the power of hydrogen ions”) or “negative logarithm” (referring to the mathematical description of hydrogen ion concentration).
—Cherie Winner




