A New Way to Catch the Rain
The carbon budget of the upper ocean includes an important loss to the deep ocean due to a very slowly falling rain of organic particles, usually called sediment. As this sediment falls through the upper water column it is consumed, mainly by bacteria, and the carbon is recycled into nonsinking forms (dissolved or colloidal organic carbon or inorganic forms). Thus the sediment rain decreases with increasing depth in the water column, and only a tiny fraction reaches the deep sea floor, less than about one percent.
December 1997 — The carbon budget of the upper ocean includes an important loss to the deep ocean due to a very slowly falling rain of organic particles, usually called sediment. As this sediment falls through the upper water column it is consumed, mainly by bacteria, and the carbon is recycled into nonsinking forms (dissolved or colloidal organic carbon or inorganic forms). Thus the sediment rain decreases with increasing depth in the water column, and only a tiny fraction reaches the deep sea floor, less than about one percent.
It is of great interest to observe the sediment rain at different depths in the ocean so that the recycling rate can be quantified. There is a long (and contentious) history of sample collection in simple, open-topped cylinders or “traps.” For reasons of technical convenience, these traps have been either attached to moorings or tethered from surface floats. The ongoing Bermuda Atlantic Time Series (BATS) program utilizes surface-tethered traps to catch sediment falling through the upper 300 meters of the ocean. These traps are deployed for three to five days during the regular monthly BATS cruises, and the contents are then analyzed for the carbon, nitrogen, and other elements being carried to the deep ocean by the sediment rain. BATS investigators, especially Tony Michaels, now at the University of Southern California’s Wrigley Institute, who first interested us in this problem, have been unable to close the upper ocean carbon budget using these data. It seems that on annual average, there is an unaccounted loss in the carbon budget. Perhaps there is an important unknown process acting to deplete the upper ocean carbon budget, or perhaps the surface-tethered sediment traps may be undercollecting the sediment. The former would be the most exciting result, but there is good reason to suspect a role for collection error, and that is what we have begun to address.
A collection error by a moored or a surface-tethered sediment trap is easy to imagine; the sediment falls through the water at a rate of about 10 to 200 meters per day, while it is carried horizontally by currents at a rate of from 5 to 50 kilometers per day (typical of currents in the upper ocean-deep ocean currents may be much less). A fixed or surface-tethered trap will thus be immersed in a nearly horizontal flux of sediment. Flow past the trap would set up vortices that could either enhance or reduce the collection of sediment, depending upon the fall speed of the sediment, the speed of the current, and the tilt and geometry of the trap. Similar collection problems are known to afflict most rain gauges (we mean water rain!), and the phenomenon can be reproduced and studied in controlled, laboratory experiments. But even if we knew in great detail the dynamics of this collection error, we could not apply this knowledge to correct the presently measured sediment flux since the currents and the sediment falling speeds are highly variable and, in practice, not easily measured.
An obvious solution is to make a sediment trap that drifts freely with the currents, so that the flow past the trap is effectively zero (imagine the wind blowing past a hot air balloon). Based upon this idea, and armed with a grant from the Green Foundation, we have developed and recently begun to test a new sediment trap that we call the Neutrally Buoyant Sediment Trap (NBST). The NBST is built from components and techniques familiar to us from our work with a variety of neutrally buoyant float systems. The main challenge for the NBST was to float at a prescribed depth, with shallower depths, 150 to 300 meters, being the most difficult. Around Bermuda, where we have started NBST testing, the ocean is very weakly stratified at these depths, and thus the ballasting of an NBST becomes extraordinarily sensitive. An error of just one gram in the weight of a 16 kilogram instrument leads to a depth error of about 40 meters, which is unacceptable. The NBST traps also carry a significant load of denser water (an unpleasant solution of saline and formaldehyde to preserve the samples and discourage theft by midwater scavengers, mainly small shrimp) that can be partially flushed out during launch. To overcome these ballasting issues we decided to endow the NBST with a variable displacment device (a cylinder and piston in contact with the sea) and a very modest brain, or microprocessor, so that it could be self-ballasting. Thus the NBST measures pressure, and if it finds that it is deeper, say, than its target depth, then it increases its displacement by forcing the piston outwards, something like a miniature submarine blowing ballast. By repeated checking and correcting at hourly intervals, the NBST can be made to float at a prescribed depth almost anywhere in the water column.
While the NBST idea is inherently simple, it still happens that testing the prototype NBSTs at sea has been hair raising, since they are autonomous and not command-recoverable once
they are launched. On our first field test in summer 1996 we deployed a glass-hulled instrument (glass has several important advantages as a hull material, including being able to endure very high pressures). Unfortunately, the sea state was high at the time of deployment, and we bumped the NBST with the ship. This cracked the hull, and led to a prompt sinking (we now make the hulls from aluminum tubing that is better able to take such abuse). On a later trial we lost two out of two instruments due, we think, to electrical interference between a new high voltage light, intended to be used as a recovery beacon, and the microprocessor (we have redesigned the light).
These failures were a loss of time and resources, and they were also intensely disappointing. But we were confident that our basic idea had merit, and we persisted in building and deploying NBST prototypes until finally achieving real success during this past summer and fall. With assistance from Debbie Steinberg and co-workers at the Bermuda Biological Station for Research, we obtained our first NBST samples during the summer of 1997. During this test an NBST at 150 meters collected approximately the same sediment flux as a surface-tethered trap at the same depth; however, a second NBST at 250 meters depth collected significantly more material than the comparable surface-tethered trap. Thus the profile of the sediment flux, which is a direct consequence of the recycling process, appears very different when measured by an NBST compared with conventional traps (the NBST-measured flux appears to fall off less rapidly with depth). On still another successful deployment this past fall, an NBST at 150 meters collected significantly less material than did a comparable surface-tethered trap, and we found that the kind and quality of the collected material was markedly different.
Our story of upper ocean sediment trapping is still unfolding, but we can already see that it will not be as straightforward as a simple under- or over-collection error. If indeed there is a collection error by surface-tethered traps (and there probably is, given our results to date), then it likely depends upon the season, since the kind of material that makes up the sediment flux changes with season, and also with depth. During the next two years we hope to make a series of comparisons over a full annual cycle as part of the BATS program. These new data are sure to spark a great deal of interest among the geochemists who have grappled with these difficult problems, and it may, perhaps, inspire other engineers to conceive still better means to measure this very gentle but crucially important rain of organic material that falls through the upper oceans.
The authors are grateful to the Green Foundation for a technology development grant that made their floating sediment trap project possible.
Authors Valdes, Buesseler, and Price collectively have 58 years of service to WHOI science (23, 17, and 18 years, respectively), including many months at sea for each. Buesseler, who is a 1987 graduate of the MIT/WHOI Joint Program is currently in Arlington, VA, at the National Science Foundation, where he is Associate Program Director in Chemical Oceanography.
Slideshow

Slideshow
 Jim Price (left) and Jim Valdes with a prototype of the neutrally buoyant sediment trap. The four small plastic tubes collect sediment and the large central tube houses electronics, a variable displacement device, recovery beacons, and batteries.
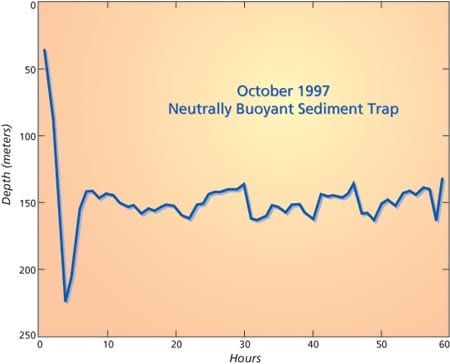
Jim Price (left) and Jim Valdes with a prototype of the neutrally buoyant sediment trap. The four small plastic tubes collect sediment and the large central tube houses electronics, a variable displacement device, recovery beacons, and batteries.- A record of pressure measured by a neutrally buoyant sediment trap during a three day-long deployment. The trap is passive for the first three hours after launch, and then begins to check and correct depth at hourly intervals. This instrument was targeted for 150 meters depth, and was slightly heavy as it was launched. At hour four it rose to the target depth by increasing its displacement slightly. It then remained within about 10 meters of its target depth until the end of its mission.
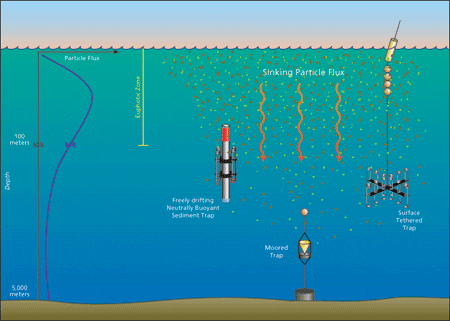
- Particle flux scientists employ three main sediment trap designs. The cylindrical, surface-tethered and free-drifting neutrally buoyant traps serve in shallow waters, and the moored conical traps are used for deep waters (drawings are not to scale). Given the generally decreased particle flux at great depths (see graph at left in the figure), the deep traps' conical shape increases the collection area--these traps have a 300 times larger collection area than the shallow water traps.