
The Secret Lives of Fish
Scientists learn to read the 'diary' recorded in the ear bones of fish
The ocean’s once-abundant fisheries—a resource that helps feed the world and drives multi-billion-dollar economies—are rapidly being depleted. Seventy percent of the ocean’s fish are being fished at or above catch limits that would sustain the fish stocks, according to a recent report by the National Research Council.
This dismal situation has led to calls for Marine Protected Areas (MPAs)—areas completely closed to fishing—as a means to protect both fish stocks and the environments they inhabit. Instead of trying to manage single species in isolation, the idea is to manage and preserve whole ecosystems.
But which areas should we protect, to protect fish stocks most effectively? To make these decisions, we need to know details about fish life cycles, movements, and migrations. Unfortunately, large gaps remain in our knowledge about the secret lives of fish.
Following fish in a vast ocean
On land, the task is much easier. To learn about movements of terrestrial animals, researchers usually do tag-recapture studies. They place tags on a number of animals, release them, and then keep track of where the tagged animals were released and where they were found at later times.
Such studies are difficult in marine environments. Larval fish, generally measuring 5 millimeters or less, are too small to tag. In addition, fish typically lay millions of eggs, of which 99.9% do not survive. Even if we could tag hatchlings, we would lose nearly all of our study subjects before they reached adulthood.
Consequently, fisheries scientists have no way to know where an adult haddock caught on Georges Bank was spawned, or the location of the nursery area where it spent its adolescence, or the likelihood that it would return as an adult to spawn in the same place. Yet, this is exactly the information about fish species that we need to select and design MPAs that will effectively conserve and replenish fish populations.
Their ears can tell tales
Our recent research points to a promising new way to reveal where and how fish live their lives. Within all fish are ear bones, called otoliths. They grow throughout each fish’s life, adding annual rings, similar to the growth rings in trees. For more than a century, biologists have used otoliths to estimate fish’s ages.
But otoliths may be able to tell us far more. Otoliths consist of alternating layers of calcium carbonate and protein, which are deposited in daily increments. Through a complicated process, the chemical composition of the calcium carbonate is influenced by the chemical composition and temperature of the water the fish inhabit. If a fish swims into waters with different chemical or physical properties, those differences will be recorded chemically in its otoliths.
In other words, the otoliths can tell us where the fish has been. And because otolith layers remain unchanged once they are deposited, they can tell us when the fish was there. In addition, in some fish species, the width of each daily growth increment in the otoliths can be correlated with the growth rate of the fish.
Keys to unlock the ‘black box’
In many ways, otoliths can be thought of as the fish-equivalent of an airplane’s flight data recorder. They are continually logging information about the growth and health of the fish and about the water it swims in. Since otoliths begin to grow just before or after hatching, the entire life history of individual fish is available to be read, albeit in code.
Unfortunately, accessing information from flight data recorders is simpler than it is from the otolith “black box.” Scientists can determine the chemical composition of samples taken from many calcium structures, such as coral skeletons or clamshells, by using a mass spectrometer. This instrument sorts individual elements within a sample according to their mass and measures the amounts of each.
But such analyses generally require fairly large amounts of material. Each day, fish deposit only an extremely thin layer of otolith—about 10 micrometers (0.0004 inches) in width. Most mass spectrometers cannot be used on such small sampling scales.
To determine the chemical composition of daily growth increments, scientists need to analyze thin (5- to 10-micrometer) sections of otoliths. To analyze these thin sections, they require special types of mass spectrometers that use microbeams of ions or laser probes.
Scientists are fortunate to have access to such state-of-the-art mass spectrometers, including the Northeast National Ion Microprobe Facility (NNIMF) and the Plasma Induced Multi-Collector Mass Spectrometer (PIMMS) facility, located at Woods Hole Oceanographic Institution. These provide precise measurements of minute quantities of trace elements and isotopes in thin sections of the otoliths. These measurements give us the ability to discern small differences in chemical composition that occur within time periods as short as days.
Cracking the chemical code
Once collected, the data are still difficult to interpret, however. When otoliths form, they are surrounded by the fish’s internal fluids. These fluids are separated from the ambient water on the other side of the fish’s scales. So the possibility has existed that otolith chemistry has no relationship to the chemistry of the ambient seawater outside the fish.
Our research shows evidence, however, that chemistry of the water the fish swims in does indeed influence the chemical composition of its otoliths. We demonstrated in the laboratory that for at least two elements, barium and strontium, there is a direct, linear relationship between concentrations of these elements in the ambient water and in the otoliths. This may hold true for other elements, too.
If the properties of ambient water do influence the chemical composition of the otoliths on a daily basis, can we use the variations in composition as natural records of a fish’s hatching location and subsequent travels?
A treasure trove of fish data
We have recently shown that we can do so with a natural, wild population of weakfish (Cynoscion regalis). Currently, these fish are managed as if they are a single population along the whole U.S. East Coast. That is because weakfish living from Florida to Maine show no genetic differences. Weakfish are an important commercial and recreational species that hatch in estuaries, spend their adulthood near the bottom in coastal waters, and return to estuaries to spawn.
Juvenile weakfish, however, hatch in each of five different East Coast estuaries. They are Doboy Sound, Georgia; Pamlico Sound, North Carolina, Chesapeake Bay, Virginia; Delaware Bay, Delaware; and Peconic Bay, near the west end of Long Island, New York.
We have found that otoliths of fish born in each of the five natal estuaries had different, unique isotope and element compositions, or “signatures.” All their lives, these fish had carried a natural tag, encoding the location where they were hatched.
We then analyzed otolith cores (the first portions deposited by hatchlings) from adult fish in those estuaries, and we found that most of the adult fish were returning to their birthplaces to reproduce—not randomly to any of the five possible natal estuaries. Knowing this means that protecting just one or two natal estuaries might not be sufficient to maintain the fish stocks.
We now believe that fish otoliths are a rich source of demographic information for fisheries scientists all over the world. At least one million otoliths are sectioned in laboratories every year, primarily to determine the fish’s ages. Now we know that annual and daily growth increments in otoliths contain significantly more information about the secret lives of fish than simply their age. Chemical signatures in the otoliths offer the potential to reveal where and when a fish traveled throughout its life.
The development of techniques for decoding this otolith archive gives us a powerful new tool to help manage fisheries resources. If we know where fish hatch and travel, and where the spawning adults originate, fisheries managers will be better able to choose the most effective locations to site MPAs and to restrict fishing—to protect the world’s diminishing fish resources.
Slideshow

Slideshow
 Simon Thorrold examines a magnified otolith (ear bone) of a weakfish. Dark and light lines are alternating layers of calcium carbonate and protein, secreted as layers that can be detected as annual, or even daily, rings. (Photo by Tom Kleindinst, WHOI)
Simon Thorrold examines a magnified otolith (ear bone) of a weakfish. Dark and light lines are alternating layers of calcium carbonate and protein, secreted as layers that can be detected as annual, or even daily, rings. (Photo by Tom Kleindinst, WHOI)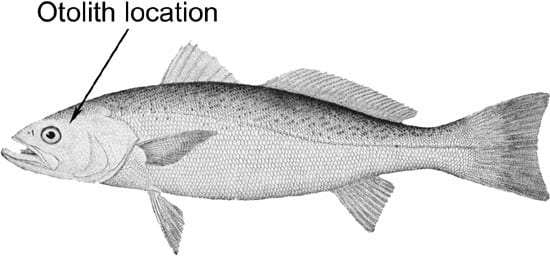 The weakfish, Cynoscion regalis
The weakfish, Cynoscion regalis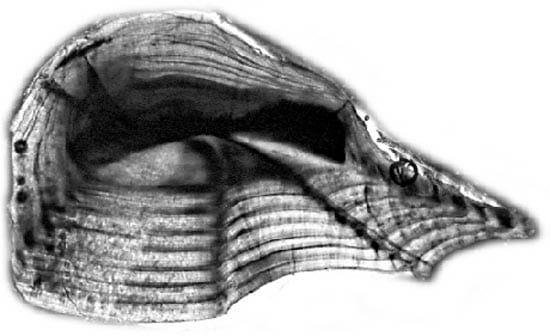 Alternating light and dark bands of calcium carbonate and protein are visible in this enlarged picture of a weakfish otolith, taken through a light microscope. (Photo by Simon Thorrold, WHOI)
Alternating light and dark bands of calcium carbonate and protein are visible in this enlarged picture of a weakfish otolith, taken through a light microscope. (Photo by Simon Thorrold, WHOI)
(Simon Thorrold, WHOI.)
Related Articles
See Also
- Simon Thorrold's Lab Site
- In Tiny Ear Bones, the Life Story of a Giant Bluefin Tuna from Oceanus magazine
- Tracking Fish to Save Them from Oceanus magazine
- Thorrold's Fish Research
- Science Magazine: Ear Bones Reveal Homing Tendencies
