Dr. John B. Waterbury
Scientist Emeritus
Biology
Contact Information:
Work: 508-289-2742
jwaterbury@whoi.edu
Building: Watson Building 107
Mailing Address:
266 Woods Hole Road, MS #51
Woods Hole Oceanographic Institution
Woods Hole, MA 02543
Research Interests
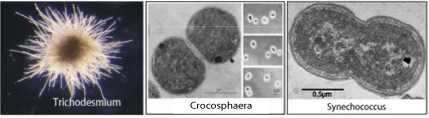 Cyanobacteria, including species of Synechococcus, Trichodesmium, and Crocosphaera, are prominent constituents of the marine biosphere that account for a significant percentage of global primary productivity. Additionally, in warm waters diazotrophic cyanobacteria (i.e., Trichodesmium and Crocosphaera) are vital components of the global nitrogen cycle through the production of “new” nitrogen. By studying the physiology of these important microorganisms at the cellular and molecular levels, we will gain new insights into the factors that affect their growth in situ and greatly increase our understanding of how marine nutrient cycles impact global processes.
Cyanobacteria, including species of Synechococcus, Trichodesmium, and Crocosphaera, are prominent constituents of the marine biosphere that account for a significant percentage of global primary productivity. Additionally, in warm waters diazotrophic cyanobacteria (i.e., Trichodesmium and Crocosphaera) are vital components of the global nitrogen cycle through the production of “new” nitrogen. By studying the physiology of these important microorganisms at the cellular and molecular levels, we will gain new insights into the factors that affect their growth in situ and greatly increase our understanding of how marine nutrient cycles impact global processes.
Education
B.A., University of Vermont, 1965
M.A., University of California, Berkeley, 1971
Ph.D., University of California, Berkeley, 1976
Awards
- Elected Fellow of the American Association for the Advancement of Science, 1994
- Elected Fellow of the American Academy of Microbiology, 2003
- Recipient of the 2012 Gilbert Morgan Smith Medal from the National Academy of Science
Oceanus Magazine
Article by
- Little Things Matter a Lot
December 2004 - Creature Feature: Viruses of Marine Bacteria
Vol. 35, No. 3
Featured in
- A Green Thumb for Ocean Microbes
Winter 2016 - Recycling Rare, Essential Nutrients in the Sea
January 2011 - Tracking Nitrogen’s Elusive Trail in the Ocean
December 2008 - A Most Ingenious Paradoxical Plankton
December 2008
